Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shaanxi click biotechnology co., Ltd | China | |||
|---|---|---|---|---|
 | www.herbchemical.com | |||
 | +86 18629008060 | |||
 | anna@herbchemical.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Cheng Du Chenlv Herb Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chenlvherbs.com | |||
 | +86 18981728582 | |||
 | chenlvherb@126.com | |||
 | WeChat: Cheng Du Chenlv Herb Co., Ltd. | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Alsachim SAS | France | |||
|---|---|---|---|---|
 | www.alsachim.com | |||
 | +33 (0) 390 402 200 | |||
 | +33 (0) 390 402 199 | |||
 | contact@alsachim.com | |||
| Chemical manufacturer | ||||
| Natland International Corporation | USA | |||
|---|---|---|---|---|
 | www.natland.com | |||
 | +1 (919) 380-9775 | |||
 | +1 (919) 380-9886 | |||
 | info@natland.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | (+/-)-Naringenin |
| Synonyms | 2,3-Dihydro-5,7-dihydroxy-2-(4-hydroxyphenyl)-4H-1-benzopyran-4-one; 4',5,7-Trihydroxyflavanone; 5,7,4'-Trihydroxyflavanone |
| Molecular Structure | 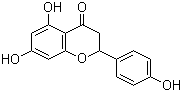 |
| Molecular Formula | C15H12O5 |
| Molecular Weight | 272.25 |
| CAS Registry Number | 67604-48-2 |
| EC Number | 266-769-1 |
| SMILES | C1C(OC2=CC(=CC(=C2C1=O)O)O)C3=CC=C(C=C3)O |
| Density | 1.5±0.1 g/cm3 Calc.*, 1.485 g/mL (Expl.) |
|---|---|
| Melting point | 247 - 250 °C (Expl.) |
| Boiling point | 577.5±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 224.7±23.6 °C (Calc.)* |
| Index of refraction | 1.693 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(+/-)-Naringenin is a flavanone, a subclass of flavonoids, widely found in citrus fruits such as grapefruits, oranges, and lemons. Chemically, it is characterized by a 2,3-dihydro-2-phenylchromen-4-one skeleton with hydroxyl groups at the 5, 7, and 4′ positions, and the racemic mixture designation (+/-) indicates it contains both enantiomers. Naringenin has drawn significant scientific attention due to its diverse pharmacological activities, including antioxidant, anti-inflammatory, hepatoprotective, anticancer, and cardioprotective effects. The discovery of naringenin originated from phytochemical investigations into citrus fruits in the early 20th century, aimed at identifying bioactive constituents responsible for the fruits’ health-promoting properties. Through solvent extraction, chromatography, and spectroscopic analysis, researchers isolated and characterized naringenin, enabling studies on its biological activities and mechanisms of action. Pharmacologically, naringenin exhibits potent antioxidant activity. Its phenolic hydroxyl groups allow it to scavenge reactive oxygen species (ROS), inhibit lipid peroxidation, and protect cellular biomolecules from oxidative damage. This activity underpins its protective effects in tissues subjected to oxidative stress, including the liver, cardiovascular system, and neuronal cells. Naringenin also demonstrates anti-inflammatory properties. It can suppress the production of pro-inflammatory mediators, including tumor necrosis factor-α (TNF-α) and interleukins, and inhibit enzymes such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS). These effects are mediated through modulation of signaling pathways such as NF-κB and MAPK, providing potential therapeutic applications in chronic inflammatory conditions. Cardioprotective effects of naringenin have been observed in both preclinical and clinical studies. It can improve endothelial function, reduce oxidative stress in vascular tissues, and inhibit platelet aggregation, which may lower the risk of atherosclerosis and other cardiovascular diseases. Hepatoprotective effects are also notable; naringenin can reduce liver injury induced by toxins or metabolic stress, likely due to its antioxidant and anti-inflammatory mechanisms. Naringenin exhibits anticancer activity through multiple mechanisms, including induction of apoptosis, inhibition of cell proliferation, and cell cycle arrest in various cancer cell lines. Its effects have been documented in cancers of the breast, liver, colon, and prostate, and are mediated via signaling pathways such as PI3K/Akt, p53, and MAPK. Despite its broad pharmacological profile, naringenin has limited water solubility and bioavailability, which restricts its systemic absorption. Various formulation strategies, including nanoparticles, liposomal encapsulation, and cyclodextrin inclusion complexes, have been explored to improve solubility, stability, and bioavailability, enhancing its therapeutic potential. In addition to therapeutic applications, naringenin is used as a dietary supplement due to its natural occurrence in citrus fruits and its recognized health benefits. Its safety profile is favorable, with minimal adverse effects reported in both preclinical and clinical studies. Overall, (+/-)-naringenin is a bioactive flavanone with antioxidant, anti-inflammatory, hepatoprotective, cardioprotective, and anticancer properties. Its isolation from citrus fruits and subsequent pharmacological studies highlight its potential as a natural therapeutic agent, and ongoing research continues to explore its molecular mechanisms, bioavailability enhancements, and clinical applications. References 2010. Preparation and physicochemical properties of the complex of naringenin with hydroxypropyl-beta-cyclodextrin. Molecules (Basel, Switzerland). DOI: 10.3390/molecules15064401 2010. Suppression of hepatic oxidative events and regulation of eNOS expression in the liver by naringenin in fructose-administered rats. European Journal of Pharmacology. DOI: 10.1016/j.ejphar.2010.07.015 2008. The citrus fruit flavonoid naringenin suppresses hepatic glucose production from Fao hepatoma cells. Molecular Nutrition & Food Research. DOI: 10.1002/mnfr.200700514 |
| Market Analysis Reports |