Online Database of Chemicals from Around the World
| Zibo Aote Fine Chemical Factory | China | |||
|---|---|---|---|---|
 | www.aotechem.com | |||
 | +86 (533) 758-0220 +86 13969356115 | |||
 | +86 (533) 752-1583 | |||
 | sales@aotechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Chemtech Inductry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjchemtech.com | |||
 | +86 (571) 5671-5668 | |||
 | +86 (571) 85423167 | |||
 | info@zjchemtech.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Qinba Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shangfluoro.com | |||
 | +86 (21) 6517-0832 +86 17301729617 | |||
 | +86 (21) 6517-0832 | |||
 | shangfluoro@hotmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2017 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon halide |
|---|---|
| Name | Heptafluoroisopropyl iodide |
| Synonyms | Heptafluoro-2-iodopropane; Perfluoro-2-iodopropane; 1,1,1,2,3,3,3-heptafluoro-2-iodo-propane |
| Molecular Structure | 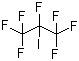 |
| Molecular Formula | C3F7I |
| Molecular Weight | 295.93 |
| CAS Registry Number | 677-69-0 |
| EC Number | 211-643-3 |
| SMILES | C(C(F)(F)F)(C(F)(F)F)(F)I |
| Density | 2.096 |
|---|---|
| Melting point | -58 °C |
| Boiling point | 39 °C |
| Refractive index | 1.327-1.33 |
| Water solubility | insoluble |
| Hazard Symbols |     GHS06;GHS07;GHS08;GHS09 Danger Details GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H331-H331-H335-H372-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P302+P352-P304+P340-P305+P351+P338-P316-P317-P319-P321-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Heptafluoroisopropyl iodide, with the chemical formula C3HF7I, is an organofluorine compound that has garnered attention due to its unique chemical properties and potential applications. The discovery of heptafluoroisopropyl iodide can be traced back to advancements in the field of organofluorine chemistry in the late 20th century. Fluorinated compounds have long been of interest to chemists due to the distinctive properties imparted by the presence of fluorine atoms, such as increased stability and enhanced reactivity. Heptafluoroisopropyl iodide is synthesized through various methods, including the reaction of heptafluoroisopropanol with iodine or iodine-containing reagents. The introduction of iodine into the heptafluoroisopropyl group creates a compound that exhibits both hydrophobic and lipophobic characteristics, making it useful in specialized applications. One of the primary applications of heptafluoroisopropyl iodide is in the field of pharmaceuticals and agrochemicals, where it serves as a valuable intermediate in the synthesis of more complex fluorinated compounds. Fluorinated organic molecules are widely recognized for their enhanced biological activity and metabolic stability, which makes them attractive candidates for drug development and agricultural formulations. Additionally, heptafluoroisopropyl iodide is utilized in the development of advanced materials, particularly in the manufacturing of coatings and surfactants. Its unique properties enable the creation of hydrophobic surfaces that repel water, oils, and other contaminants, making it suitable for applications in protective coatings, stain-resistant textiles, and other innovative materials. The compound has also attracted attention in the realm of green chemistry due to its potential as a solvent in various chemical reactions. Solvents play a crucial role in determining the efficiency and selectivity of chemical processes, and the unique properties of heptafluoroisopropyl iodide may provide advantages over traditional organic solvents. Despite its promising applications, research on heptafluoroisopropyl iodide is still emerging, and there are ongoing studies to further explore its reactivity and potential uses. As with many fluorinated compounds, considerations regarding environmental impact and safety are essential. The persistence and potential toxicity of fluorinated substances require careful handling and regulation to mitigate any adverse effects on health and the environment. In conclusion, heptafluoroisopropyl iodide represents a significant advancement in organofluorine chemistry, with various applications in pharmaceuticals, agrochemicals, and advanced materials. As research continues to uncover its full potential, heptafluoroisopropyl iodide is positioned as a valuable compound in the development of innovative technologies and sustainable practices within the chemical industry. References 2017. Cobalt-Catalyzed Perfluoroalkylation of Quinoline Amides at the C5 Position. Synthesis, 49(11). DOI: 10.1055/s-0036-1588160 2005. Isotope Effects and Final Products in Pyrolysis of Halomethanes and Perfluoroalkyl Iodides. Russian Journal of General Chemistry, 75(9). DOI: 10.1007/s11176-005-0439-z |
| Market Analysis Reports |