Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | 5-Acetyl-2-Thiophenecarboxamide |
| Molecular Structure | 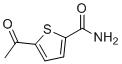 |
| Molecular Formula | C7H20NO2S |
| Molecular Weight | 169.20 |
| CAS Registry Number | 68257-89-6 |
| EC Number | 614-390-0 |
| SMILES | CC(=O)C1=CC=C(S1)C(=O)N |
| Solubility | Slightly Soluble (1.3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.310±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.590, Calc.* |
| Boiling Point | 359.3±27.0 °C (760 mmHg), Calc.* |
| Flash Point | 171.1±23.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P312+P330 Details |
| SDS | Available |
|
5-Acetyl-2-thiophenecarboxamide is an organic compound that consists of a thiophene ring, an acetyl group, and a carboxamide group. The compound is part of a larger class of substituted thiophenes, which are known for their diverse biological activities and usefulness in organic synthesis. Thiophene derivatives have garnered attention due to their unique electronic properties and their ability to participate in various chemical reactions, making them valuable intermediates in pharmaceutical and materials chemistry. The discovery of 5-acetyl-2-thiophenecarboxamide can be attributed to the ongoing exploration of thiophene derivatives with modified functional groups. In the case of 5-acetyl-2-thiophenecarboxamide, the acetyl group at the 5-position and the carboxamide group at the 2-position of the thiophene ring provide enhanced reactivity compared to the parent thiophene structure. This modification opens up a range of synthetic possibilities, including the introduction of other functional groups that can modify the compound’s physical and chemical properties. In terms of its applications, 5-acetyl-2-thiophenecarboxamide has found utility primarily in the field of organic synthesis. The functional groups attached to the thiophene ring make this compound an important intermediate in the creation of more complex molecules, including those with potential pharmaceutical properties. The compound’s ability to undergo various chemical transformations, such as nucleophilic substitution and electrophilic aromatic substitution, makes it a valuable building block for the design of biologically active molecules. One area where 5-acetyl-2-thiophenecarboxamide shows promise is in the development of new drugs. Thiophene derivatives are often explored for their antimicrobial, anti-inflammatory, and anticancer activities. The acetyl group in this compound can be reactive, allowing for the modification of its biological activity, while the carboxamide group could participate in hydrogen bonding and other molecular interactions that are critical for binding to biological targets. These properties suggest that 5-acetyl-2-thiophenecarboxamide may serve as a lead compound for the design of novel therapeutic agents. Additionally, 5-acetyl-2-thiophenecarboxamide could have applications in materials science. Thiophene derivatives are commonly used in the synthesis of conductive polymers, which have potential uses in electronic devices such as organic light-emitting diodes (OLEDs), organic solar cells, and field-effect transistors. The presence of the acetyl and carboxamide groups could impart solubility, stability, and processability to polymers made from this compound, making it suitable for inclusion in electronic materials. References none |
| Market Analysis Reports |