Online Database of Chemicals from Around the World
| Earlier HK Company Limited | China | |||
|---|---|---|---|---|
 | www.earlierchem.com | |||
 | +86 (10) 6142-6769 +86 18616907430 | |||
 | +86 (10) 6142-6769 | |||
 | info@earlierchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Premium supplier since 2014 | ||||
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | N,N'-Diisopropylcarbodiimide |
| Synonyms | N,N'-Methanetetraylbis(1-methylethylamine); Dipcdi; DIC; N,N'-Methanetetraylbis(2-propanamine) |
| Molecular Structure | 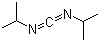 |
| Molecular Formula | C7H14N2 |
| Molecular Weight | 126.20 |
| CAS Registry Number | 693-13-0 |
| EC Number | 211-743-7 |
| SMILES | CC(C)N=C=NC(C)C |
| Density | 0.806 |
|---|---|
| Boiling point | 145-148 °C |
| Refractive index | 1.432-1.435 |
| Flash point | 33 °C |
| Hazard Symbols |       GHS02;GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details GHS02;GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H315-H317-H318-H330-H334-H335-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P271-P272-P273-P280-P284-P302+P352-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P320-P321-P332+P317-P333+P317-P342+P316-P362+P364-P370+P378-P391-P403-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
N,N'-Diisopropylcarbodiimide (DIC) is a valuable reagent used extensively in organic synthesis, particularly in peptide coupling and amide bond formation. Its unique properties and reactivity make it a staple in both academic research and industrial applications. N,N'-Diisopropylcarbodiimide was first synthesized in the mid-20th century, when chemists explored the potential of various carbodiimides in promoting chemical reactions. DIC, with the chemical formula C₇H₁₄N₂, is a colorless to pale yellow liquid with a strong and distinctive odor. It is soluble in many organic solvents, including dichloromethane and tetrahydrofuran. The structure of DIC consists of two isopropyl groups attached to the nitrogen atom of the carbodiimide functional group (−N=C=N−). This arrangement produces significant steric hindrance, which affects its reactivity and selectivity in various chemical processes. One of the main uses of DIC is in peptide synthesis. DIC acts as a coupling agent, promoting the formation of amide bonds between amino acids. It reacts with carboxylic acids and amines to form stable peptide bonds, making it essential in peptide and protein synthesis. In addition to peptide synthesis, DIC is used to form amide bonds in a variety of organic molecules. This application is crucial in the synthesis of pharmaceuticals, agrochemicals, and natural products, as amide bonds are a common structural feature of these products. DIC also promotes esterification reactions, helping carboxylic acids and alcohols to form esters. In addition, it is used to synthesize urea by reacting with amines. These reactions are important in the preparation of various chemical intermediates and final products. In polymer chemistry, DIC is used to make polymers with specific properties. Its ability to form strong, stable bonds makes it useful in the development of high-performance materials for coatings, adhesives, and other applications. DIC is highly reactive and can form bonds efficiently and quickly. This property is particularly useful in large-scale industrial processes where time and efficiency are critical. Reactions involving DIC are typically carried out under mild conditions, reducing the need for extreme temperatures or pressures. This makes these processes safer and more environmentally friendly. The steric hindrance provided by the isopropyl group in DIC makes the reaction highly selective, minimizing side products and increasing the yield of the desired product. Although DIC is a valuable reagent, it must be handled with care. It is flammable and may irritate the skin, eyes, and respiratory system. Appropriate safety measures include the use of personal protective equipment such as gloves, goggles, and working in a well-ventilated area. DIC should be stored in a cool, dry place away from sources of ignition and incompatible materials. References 2020. New Developments in Microwave-Assisted Solid Phase Peptide Synthesis. Methods in molecular biology (Clifton, N.J.), 2103. DOI: 10.1007/978-1-0716-0227-0_6 2013. Solid-Phase Peptide Synthesis: An Introduction. Methods in molecular biology (Clifton, N.J.), 1047. DOI: 10.1007/978-1-62703-544-6_1 2013. Linkers, Resins, and General Procedures for Solid-Phase Peptide Synthesis. Methods in molecular biology (Clifton, N.J.), 1047. DOI: 10.1007/978-1-62703-544-6_2 |
| Market Analysis Reports |