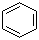
Benzene, with the chemical formula C6H6, is a fundamental aromatic hydrocarbon known for its distinct ring structure composed of six carbon atoms, each bonded to a hydrogen atom. The discovery of benzene can be traced back to 1825 when the English scientist Michael Faraday first isolated it from illuminating gas. However, it was the German chemist August Kekulé who, in 1865, proposed the cyclic structure of benzene, introducing the concept of resonance, which describes the delocalization of electrons in the ring. This structural understanding laid the foundation for modern organic chemistry.
Benzene is primarily produced through the catalytic reforming of naphtha or as a byproduct in the production of other hydrocarbons. It can also be derived from coal tar and is present in crude oil. Its widespread availability and unique chemical properties make benzene an essential building block in the synthesis of numerous organic compounds.
One of the most significant applications of benzene is as a precursor in the manufacturing of various chemicals, including ethylbenzene, cumene, and cyclohexane. Ethylbenzene is further converted into styrene, a crucial monomer for producing polystyrene, a widely used plastic. Cumene is an intermediate in the production of phenol and acetone, both of which have extensive industrial applications.
Benzene is also vital in the production of numerous pharmaceuticals and agrochemicals. It serves as a solvent in various chemical reactions and processes due to its ability to dissolve a wide range of organic compounds. In the pharmaceutical industry, benzene derivatives are utilized in the synthesis of various medications, including analgesics, antipyretics, and anti-inflammatory drugs.
Moreover, benzene plays a role in the fragrance and flavoring industries. It is used as a solvent and a carrier for various aromatic compounds, contributing to the formulation of perfumes and flavoring agents. Its pleasant odor has made it a component in several fragrance formulations, although concerns regarding its toxicity have led to stricter regulations on its use in consumer products.
Despite its numerous applications, benzene is recognized as a hazardous substance. It is classified as a carcinogen, and exposure to high levels can lead to severe health issues, including leukemia and other blood disorders. As a result, regulatory agencies have established guidelines for its handling and use in industrial settings, emphasizing the importance of safety measures to minimize exposure.
In recent years, research has focused on developing safer alternatives to benzene and finding ways to reduce its environmental impact. Green chemistry initiatives aim to create more sustainable processes for benzene production and utilization, enhancing safety while maintaining efficiency.
In conclusion, benzene is a crucial aromatic hydrocarbon with a rich history of discovery and a wide array of applications across various industries. Its role as a building block in chemical synthesis underscores its importance in modern chemistry, while ongoing research continues to address safety and environmental concerns associated with its use.
References
1979. Benzene disposition in the rat after exposure by inhalation. Toxicology and Applied Pharmacology, 49(2).
DOI: 10.1016/0041-008x(79)90441-1
1979. Dose-dependent metabolic interaction between benzene and toluene in vivo and in vitro. Toxicology and Applied Pharmacology, 48(2).
DOI: 10.1016/0041-008x(79)90030-9
1979. Service station attendants' exposure to benzene and gasoline vapors. American Industrial Hygiene Association Journal, 40(4).
DOI: 10.1080/15298667991429642
|






















































 GHS02;GHS08;GHS07 Danger Details
GHS02;GHS08;GHS07 Danger Details