Online Database of Chemicals from Around the World
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Tianjia Chemical Science & Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianjia-chem.com | |||
 | +86 (550) 702-1925 | |||
 | +86 (550) 702-1925 | |||
 | tianjiachem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Achemica | Switzerland | |||
|---|---|---|---|---|
 | www.achemica.com | |||
 | +41 (24) 466-2929 | |||
 | +41 (24) 466-2800 | |||
 | contact@achemica.com | |||
| Chemical manufacturer since 2010 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Thiazole, thiophene and pyridine |
|---|---|
| Name | 2-Amino-4,5-dimethylthiazole hydrochloride |
| Synonyms | 4,5-dimethyl-1,3-thiazol-2-amine;hydrochloride |
| Molecular Structure | 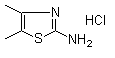 |
| Molecular Formula | C5H8N2S.HCl |
| Molecular Weight | 164.66 |
| CAS Registry Number | 71574-33-9 |
| EC Number | 633-247-3 |
| SMILES | CC1=C(SC(=N1)N)C.Cl |
| Melting point | 273-274 °C |
|---|---|
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H318-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P354+P338-P317-P319-P321-P330-P332+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-Amino-4,5-dimethylthiazole hydrochloride is an organic compound with the molecular formula C5H9ClN2S. This compound is a thiazole derivative, characterized by a five-membered ring containing both sulfur and nitrogen atoms, along with amino and methyl substituents at specific positions. The hydrochloride form is commonly used to enhance the compound's solubility in water, making it more accessible for various applications, particularly in biological and chemical research. The discovery of 2-Amino-4,5-dimethylthiazole hydrochloride is linked to the broader exploration of thiazole derivatives, which are known for their biological activity and synthetic utility. Thiazoles have been of significant interest in medicinal chemistry due to their presence in various natural products and pharmaceuticals. The specific structure of 2-Amino-4,5-dimethylthiazole, with its amino and methyl groups, gives it unique properties that can be exploited in different chemical reactions and biological studies. One of the primary applications of 2-Amino-4,5-dimethylthiazole hydrochloride is in the pharmaceutical industry, where it is used as an intermediate in the synthesis of more complex molecules. Its thiazole ring is a key structural component in many drugs, including those with antimicrobial, anti-inflammatory, and anticancer properties. The presence of an amino group at the 2-position and methyl groups at the 4- and 5-positions makes this compound particularly useful in creating molecules that can interact with biological targets, such as enzymes or receptors. In addition to its role in drug synthesis, 2-Amino-4,5-dimethylthiazole hydrochloride is also employed in chemical research as a building block for designing new compounds. Its reactive sites allow for various chemical modifications, enabling researchers to create a wide range of derivatives with potential biological or chemical activity. These derivatives can be screened for pharmaceutical applications or used in the development of new materials with specific properties. Moreover, 2-Amino-4,5-dimethylthiazole hydrochloride has been studied for its potential use in the development of dyes and pigments. The thiazole ring system, when combined with other aromatic systems, can produce compounds with interesting optical properties. These properties make such derivatives suitable for use in dyes, where color stability and intensity are important. Additionally, these compounds can be explored for their fluorescence, which is useful in imaging and diagnostic applications. Furthermore, the compound is of interest in agricultural chemistry, where thiazole derivatives are explored for their potential as agrochemicals. Compounds like 2-Amino-4,5-dimethylthiazole hydrochloride can serve as starting materials for synthesizing pesticides, herbicides, or fungicides. The structural features of the thiazole ring can be optimized to target specific pests or pathogens, contributing to more effective and sustainable agricultural practices. In summary, 2-Amino-4,5-dimethylthiazole hydrochloride is a versatile compound with significant applications in pharmaceuticals, chemical research, and materials science. Its discovery and subsequent development have led to advancements in drug synthesis, the creation of new chemical entities, and potential applications in agriculture and materials science. The compound's thiazole ring structure, combined with its functional groups, makes it a valuable tool in various scientific and industrial fields. References 2009. Identification of compounds exhibiting inhibitory activity toward the Pseudomonas tolaasii toxin tolaasin I using in silico docking calculations, NMR binding assays, and in vitro hemolytic activity assays. Bioorganic & Medicinal Chemistry Letters, 19(15). DOI: 10.1016/j.bmcl.2009.05.068 2008. Successive reaction of 2-aryl-4-dichloromethylideneoxazol-54H-ones with 2-amino-1,3-thiazoles and strongly basic nitrogen-containing reagents. Russian Journal of General Chemistry, 78(4). DOI: 10.1134/s1070363208040221 |
| Market Analysis Reports |