Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Lianyungang Jinkang Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinkang-pharma.com | |||
 | +86 (518) 8544-5517 | |||
 | +86 (518) 8544-5217 | |||
 | sales@jinkang-chem.com export@jinkang-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Trignokem International | India | |||
|---|---|---|---|---|
 | www.trignokem.com | |||
 | +91 (40) 2717-0709 | |||
 | info@trignokem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Glutamic acid derivative |
|---|---|
| Name | Sodium methotrexate |
| Synonyms | Methotrexate disodium salt; Disodium N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]-L-glutamate |
| Molecular Structure | 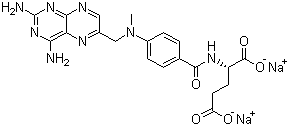 |
| Molecular Formula | C20H20N8Na2O5 |
| Molecular Weight | 498.41 |
| Protein Sequence | XE |
| CAS Registry Number | 7413-34-5 |
| EC Number | 231-022-0 |
| SMILES | CN(CC1=CN=C2C(=N1)C(=NC(=N2)N)N)C3=CC=C(C=C3)C(=O)N[C@@H](CCC(=O)[O-])C(=O)[O-].[Na+].[Na+] |
| Solubility | 2 mg/mL (water) |
|---|---|
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H315-H319-H331-H335-H340-H360 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P318-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Sodium methotrexate is a derivative of methotrexate, a well-known chemotherapeutic agent widely used in the treatment of various cancers and autoimmune diseases. It is a water-soluble salt form of methotrexate, which facilitates its administration in certain therapeutic settings. Methotrexate itself is a folic acid antagonist that inhibits the enzyme dihydrofolate reductase (DHFR), thereby interfering with the synthesis of nucleotides required for DNA synthesis. This inhibition affects rapidly dividing cells, making methotrexate an effective treatment for cancer, rheumatoid arthritis, and other diseases characterized by abnormal cell proliferation. The discovery of sodium methotrexate stems from the need to improve the solubility and bioavailability of methotrexate. While methotrexate itself is effective, its poor solubility in water can limit its clinical applications, particularly in intravenous administration. The development of sodium methotrexate was aimed at overcoming these challenges, making it easier to administer in clinical settings, especially for patients who require parenteral dosing. The sodium salt form enhances its solubility, allowing for easier formulation into injectable solutions. This modification has expanded its use, ensuring that it remains an essential part of cancer chemotherapy regimens and treatments for autoimmune diseases. Sodium methotrexate is primarily used in oncology for the treatment of various types of cancer, including leukemia, breast cancer, lung cancer, and osteosarcoma. It is also a cornerstone in the treatment of autoimmune diseases, such as rheumatoid arthritis, psoriasis, and Crohn’s disease. By inhibiting DHFR, sodium methotrexate prevents the formation of tetrahydrofolate, which is necessary for the synthesis of purines and thymidylate, components critical for DNA and RNA production. This mechanism of action makes it effective in inhibiting the growth of both cancerous cells and immune cells in autoimmune disorders. The application of sodium methotrexate extends beyond cancer and autoimmune diseases. It has been explored for its potential in treating certain viral infections, particularly in cases where the virus interferes with DNA synthesis. Additionally, sodium methotrexate is sometimes used in high-dose regimens in combination with other agents, such as leucovorin, to rescue healthy cells from the toxic effects of methotrexate. Leucovorin helps to regenerate folate in healthy cells, allowing them to survive while the cancerous cells are targeted by methotrexate. While sodium methotrexate remains a key therapeutic agent, its use is not without side effects. Common side effects include gastrointestinal issues, liver toxicity, and suppression of bone marrow function, which can lead to a higher risk of infection. Careful monitoring of patients receiving sodium methotrexate is essential to minimize these risks and to ensure the best therapeutic outcomes. References 1979. In Vivo Methotrexate Transport in Murine Lewis Lung Tumor. Journal of Pharmaceutical Sciences, 68(8). DOI: 10.1002/jps.2600680806 2024. Codelivery of metformin and methotrexate with optimized chitosan nanoparticles for synergistic triple-negative breast cancer therapy in vivo. International Journal of Pharmaceutics, 666. DOI: 10.1016/j.ijpharm.2024.124897 2025. Intradermal implantation of methotrexate-loaded puerarin-gelatin hydrogel via bubble-generating microneedles for psoriasis treatment. International Journal of Biological Macromolecules, 287. DOI: 10.1016/j.ijbiomac.2024.138201 |
| Market Analysis Reports |