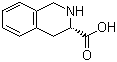
L-1,2,3,4-Tetrahydroisoquinoline-3-carboxylic acid is a chiral, partially hydrogenated heterocyclic carboxylic acid derived from the isoquinoline framework. It contains a bicyclic system composed of a benzene ring fused to a reduced pyridine ring, with a carboxylic acid substituent at the third position and an L-configuration at the stereogenic center. Compounds of the tetrahydroisoquinoline family have been known since investigations into isoquinoline reduction and related heterocycles carried out during the early development of classical organic chemistry. Studies of catalytic and chemical hydrogenation methods provided access to the tetrahydroisoquinoline core, from which carboxylated derivatives such as this compound were prepared through functional group manipulation and selective oxidation. Over time, stereoselective synthetic approaches enabled isolation of the optically pure L-enantiomer, reflecting the increased emphasis on chirality in chemical and biochemical research.
Interest in tetrahydroisoquinoline compounds increased as they were identified as structural motifs in a wide range of natural products. Alkaloids containing this scaffold were found in several plant families, especially those associated with traditional medicinal uses. This led to systematic investigation of synthetic analogues of such alkaloids to study their biological roles, metabolism, and potential therapeutic applications. The commercial development of chiral building blocks based on the tetrahydroisoquinoline skeleton followed, facilitating the incorporation of this motif into designed molecules. The carboxylic acid functionality at position three attracted attention because it permitted diverse derivatization and coupling reactions, making L-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid a versatile intermediate in both academic and industrial laboratories.
The compound has been used extensively in synthetic chemistry, particularly in the preparation of chiral amides, esters, and peptide-like molecules. The carboxyl group enables formation of amide bonds with amines, giving access to complex nitrogen-containing structures. In addition, the tetrahydroisoquinoline core serves as a foundation for rigidified ligands, pharmacophores, and stereochemically defined fragments for structure–activity studies. For this reason, it has become a frequently used intermediate in the synthesis of analogues of natural tetrahydroisoquinoline alkaloids and other nitrogen heterocycles. Hydrogenation, oxidation, and substitution reactions can be selectively applied to the core while retaining the stereochemical integrity of the L-configuration, enabling preparation of derivatives with controlled geometric and electronic properties.
In biochemical research, the compound has been applied in studies related to amino acid analogues and peptide modification. The presence of a carboxyl group adjacent to a secondary amine allows it to mimic certain structural features of amino acids while introducing a bicyclic constraint. This has made it useful in designing modified peptides where conformational restriction is necessary to probe structure–function relationships. Incorporation into peptides can alter folding, receptor binding, and metabolic stability, providing a tool for investigating the interplay between rigidity and biological activity in peptide systems. Such applications have benefited from advances in coupling procedures and protecting group strategies that maintain the integrity of the tetrahydroisoquinoline ring.
Research into stereoselective synthesis enabled development of approaches to obtain the L-isomer by asymmetric hydrogenation, enzymatic resolution, or chiral auxiliary techniques. These methods improved accessibility and reproducibility, which allowed wider adoption of the compound in laboratory practice. The ability to prepare the compound in optically enriched form was important for its use in studying interactions with enzymes, receptors, and transport systems, where enantioselectivity plays a crucial role. As studies of chirality in medicinal and biological chemistry expanded, interest in compounds such as L-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid increased correspondingly.
The compound has also been employed in methodological development in organic synthesis. It has served as a substrate in investigations of catalytic coupling, asymmetric transformation, and stereoretentive reactions. Because the bicyclic scaffold is sensitive to steric and electronic changes, it has functioned as a probe to characterize reaction pathways and selectivity trends. Additionally, its derivatives have been used to explore the impact of ring saturation, aromaticity, and substituent effects within nitrogen-containing polycyclic compounds.
Historically, the development and application of L-1,2,3,4-tetrahydroisoquinoline-3-carboxylic acid reflect broader themes in heterocyclic chemistry, including the transition from early structural studies to modern stereoselective synthesis, and from small-scale academic preparations to applied research involving pharmaceuticals and biologically active compounds. Its continuing relevance arises from its dual identity as both a recognizable natural product–related motif and a flexible, functionally rich intermediate that supports the preparation of structurally diverse molecules for chemical, biological, and medicinal research.
References
2015. Synthesis of Optically Pure (R)- and (S)-Tetrahydroisoquinoline-1- and -3-Carboxylic Acids. Synthesis, 47(7).
DOI: 10.1055/s-0034-1380289
2019. Enantioseparations by High-Performance Liquid Chromatography Based on Chiral Ligand Exchange. Methods in Molecular Biology, 1985.
DOI: 10.1007/978-1-4939-9438-0_15
|




























































































 GHS07 Warning Details
GHS07 Warning Details