Dibromoformaldoxime is a halogenated oxime derived from formaldehyde, in which two bromine atoms and an oxime functional group are attached to a one-carbon core. It belongs to a class of compounds in which carbon–nitrogen double bonds are stabilized through the presence of electron-withdrawing substituents. The compound was introduced during research into the reactivity of halooximes, a topic that developed as chemists investigated the behavior of formaldehyde derivatives and halogenated nitrogen compounds. Systematic work on oxime formation from aldehydes by reaction with hydroxylamine provided access to fundamental oxime structures, and subsequent halogenation studies led to the preparation of dibrominated analogues. As methods for controlling halogenation became more refined, dibromoformaldoxime was obtained through carefully moderated bromination in the presence of oxime functionalities.
Early investigations into its properties focused primarily on its chemical behavior under different conditions. The compound was examined for stability, modes of decomposition, and reactivity toward nucleophiles and bases. Its structure permitted researchers to examine how multiple bromine atoms influence the susceptibility of the oxime group to hydrolysis, rearrangement, or condensation. Comparisons with related chloro- and iodo-oximes revealed trends in reactivity associated with halogen size, electronegativity, and leaving-group properties. This work contributed to broader understanding of halogenated nitrogen compounds and their transformation pathways.
Interest in dibromoformaldoxime also emerged from studies of electrophilic bromination. The compound served as an illustrative example of the effect of sequential bromine substitution on a simple molecular scaffold. Researchers examined changes in melting behavior, solubility, and volatility as indicators of intermolecular interactions and crystal structure influences. Such investigations played a role in characterizing the behavior of small halogenated organic molecules and in developing purification methods appropriate for them. Fundamental studies of this type supported the later development of industrial processes using halogenated intermediates, even when a particular compound was not directly applied on a commercial scale.
From a synthetic perspective, dibromoformaldoxime has been used as a reagent and as a precursor to other brominated compounds. The bromine atoms can participate in substitution reactions, permitting conversion to organobromine derivatives under controlled conditions. The oxime group allows cyclization, reduction, and rearrangement reactions that provide access to nitrogen-containing structures. In some contexts, dibromoformaldoxime has been used to introduce both nitrogen and bromine into more complex molecules, forming intermediates relevant to heterocycle synthesis and to the preparation of functionalized building blocks. Oxime derivatives containing multiple halogens have been used to explore reaction pathways leading to nitriles, amides, and amines, and dibromoformaldoxime contributed to that body of work.
The compound has also been examined in relation to halogen transfer chemistry. Studies assessed the ability of halogenated oximes to act as donors of bromine under specific conditions, enabling controlled halogen introduction to selected substrates. Research into these reactions helped clarify the mechanistic role of oximes in halogen exchange processes and provided data relevant to the optimization of reaction selectivity. Investigators documented how solvent choice, temperature, and catalyst presence influence the course of such transformations, and dibromoformaldoxime served as one of the key test substances in this context.
Applications of dibromoformaldoxime have generally remained within specialized laboratory research. Its primary uses are associated with synthetic exploration, mechanistic studies in halogenated oxime chemistry, and preparation of derivative compounds containing both bromine and nitrogen. The compound is particularly useful in situations where a reactive brominated species bearing an oxime function is required for controlled transformations, and where the reactivity profile of bromine atoms must be studied in a simplified molecular environment. Its role in research on the chemistry of halogenated aldehyde derivatives has supported developments in analytical methodology, synthetic strategy, and understanding of structure–reactivity relationships.
The continued relevance of dibromoformaldoxime arises from its position at the intersection of halogen chemistry and oxime chemistry. It provides a compact platform on which multiple reactive features are combined, enabling systematic examination of the interplay between halogenation state and oxime behavior. As a result, the compound remains of interest in academic research and serves as a reference point for understanding the behavior of more complex halogenated nitrogen compounds.
References
2019. Lewis Acid Catalyzed Regio- and Diastereoselective Synthesis of Spiroisoxazolines via One-Pot Sequential Knoevenagel Condensation/1,3-Dipolar Cycloaddition Reaction. Synthesis, 51(4).
DOI: 10.1055/s-0037-1610676
2023. Theoretical implications on the [3 + 2] cycloaddition reactions of dibromoformaldoxime and (Z)-, (E)-3-(4-chlorobenzylidene)-1-methylindolin-2-one in terms of FMO, MEDT, and distortion-interaction theories. Structural Chemistry, 34(6).
DOI: 10.1007/s11224-023-02219-8
|































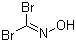

 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details