Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Chemtech Inductry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjchemtech.com | |||
 | +86 (571) 5671-5668 | |||
 | +86 (571) 85423167 | |||
 | info@zjchemtech.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Thermo Fisher Scientific | USA | |||
|---|---|---|---|---|
 | www.fishersci.com | |||
 | +1 (800) 766-7000 | |||
 | +1 (800) 926-1166 | |||
 | fs.order@thermofisher.com | |||
| Chemical manufacturer | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Simple substance |
|---|---|
| Name | Litium |
| Synonyms | Lithium metal |
| Molecular Structure | 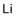 |
| Molecular Formula | Li |
| Molecular Weight | 6.94 |
| CAS Registry Number | 7439-93-2 |
| EC Number | 231-102-5 |
| SMILES | [Li] |
| Density | 0.534 g/mL |
|---|---|
| Melting point | 180 °C |
| Boiling point | 1340 °C |
| Water solubility | REACTS |
| Hazard Symbols |   GHS02;GHS05 Danger Details GHS02;GHS05 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H260-H314 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P223-P231+P232-P260-P264-P264+P265-P280-P301+P330+P331-P302+P335+P334-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P370+P378-P402+P404-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 1415 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Lithium, with the symbol Li and atomic number 3, is a soft, silvery-white alkali metal that belongs to Group 1 of the periodic table. Its discovery can be traced back to 1817 when the Swedish chemist Johan August Arfwedson first identified it in the mineral petalite (LiAlSi4O10) while analyzing the mineral's composition. Although lithium was discovered in a mineral, it was not until 1855 that it was isolated in its elemental form by the English chemist Richard Laming through electrolysis of lithium chloride. Since its discovery, lithium has garnered significant attention for its unique properties and applications. Lithium is known for its high reactivity and low density, which makes it the lightest metal and the least dense solid element. Its ability to form stable compounds with other elements is a key feature that has facilitated its diverse applications. One of the most notable uses of lithium is in the production of lithium-ion batteries, which are widely utilized in portable electronic devices, electric vehicles, and renewable energy storage systems. Lithium-ion batteries have revolutionized the energy storage industry due to their high energy density, lightweight nature, and ability to be recharged multiple times. In addition to its role in batteries, lithium is used in various other applications. It is a critical component in the production of heat-resistant glass and ceramics, enhancing the durability and thermal stability of these materials. Lithium compounds, such as lithium carbonate, are utilized in the manufacture of specialized lubricating greases, which are employed in high-temperature and high-pressure environments, such as in aerospace and automotive industries. Furthermore, lithium has significant applications in the pharmaceutical field, particularly in the treatment of bipolar disorder. Lithium salts, such as lithium carbonate, have mood-stabilizing effects and are prescribed to manage the manic and depressive episodes associated with this condition. This therapeutic use of lithium is well-documented, and it remains one of the primary treatments for bipolar disorder, demonstrating its importance in mental health care. The demand for lithium has surged in recent years, driven primarily by the growth of the electric vehicle market and the push for renewable energy solutions. As countries and manufacturers aim to reduce carbon emissions and transition to more sustainable energy sources, lithium's role as a key ingredient in battery technology has become increasingly vital. This surge in demand has prompted extensive exploration for lithium resources, including mining operations in regions such as Australia, South America, and China. Despite its beneficial applications, the extraction and processing of lithium raise environmental concerns. Lithium mining can lead to habitat destruction, water shortages, and pollution, prompting a growing interest in sustainable practices within the industry. Efforts are underway to develop more environmentally friendly methods of lithium extraction and to recycle lithium from used batteries to reduce the need for new mining operations. In summary, lithium is a versatile alkali metal with a rich history of discovery and significant applications across various fields, particularly in energy storage, materials science, and pharmaceuticals. Its importance in modern technology continues to grow, highlighting the need for sustainable practices to ensure its availability for future generations. References 1979. Lithium fluxes in human erythrocytes. American Journal of Physiology-Cell Physiology, 237(1). DOI: 10.1152/ajpcell.1979.237.1.c102 2024. A physical perspective on lithium therapy. Progress in Biophysics and Molecular Biology. DOI: 10.1016/j.pbiomolbio.2024.11.002 2024. Continuous Monitoring of Lithium Ions in Lithium-Rich Brine Using Ion Selective Electrode Sensors Modified with Polyelectrolyte Multilayers of Poly(allylamine hydrochloride)/Poly(sodium 4-styrenesulfonate). Environmental Science & Technology. DOI: 10.1021/acs.est.4c07155 |
| Market Analysis Reports |