Lead(II) sulfate is an inorganic compound with the chemical formula PbSO4. It consists of a lead(II) cation (Pb2+) and a sulfate anion (SO42–), forming a white crystalline solid that is insoluble in water. Lead(II) sulfate is commonly found as a naturally occurring mineral called anglesite and is an important compound in both industrial and electrochemical applications.
The history of lead(II) sulfate dates back to early studies of lead salts in the 18th and 19th centuries. Chemists observed its formation when lead-containing materials were exposed to sulfuric acid or sulfates. Its low solubility and chemical stability made it useful in various chemical processes and analytical applications.
One of the most significant applications of lead(II) sulfate is in the lead-acid battery, where it forms on both the positive and negative electrodes during battery discharge. In this electrochemical process, lead dioxide (PbO2) and spongy lead react with sulfuric acid to produce lead(II) sulfate and water, releasing electrical energy. During charging, the lead(II) sulfate is converted back to lead dioxide and metallic lead, allowing the battery to be reused. The formation and reconversion of PbSO4 are central to the operation of lead-acid batteries, which remain widely used in automotive and backup power applications.
Lead(II) sulfate is also employed in the chemical industry as a precursor for the preparation of other lead compounds. It reacts with strong acids or bases to form lead salts with varying solubility and reactivity. Historically, it has been used in pigments, though this application has decreased due to toxicity concerns.
The compound can be synthesized by directly reacting lead(II) nitrate or lead(II) acetate with sulfuric acid, resulting in precipitation of PbSO4. Its low solubility in water allows for easy isolation as a solid. Safety precautions are critical because lead compounds are highly toxic, and exposure can cause severe health effects, including neurological and renal damage. Proper handling, storage, and disposal are strictly required in laboratory and industrial settings.
Lead(II) sulfate exemplifies a chemically stable, low-solubility lead salt with vital industrial applications, particularly in energy storage, and serves as a classical example of the interplay between inorganic chemistry and electrochemistry.
References
2019. Lead immobilization assisted by fungal decomposition of organophosphate under various pH values. Scientific Reports, 9.
DOI: 10.1038/s41598-019-49976-3
2014. Lead sulfate nano- and microparticles in the acid plant blow-down generated at the sulfuric acid plant of the El Teniente mine, Chile. Environmental science. Processes & impacts, 16(12).
DOI: 10.1039/c4em00483c
2005. Low temperature catalytic conversion of methane to methanol by barium sulfate nanotubes supporting sulfates: Pt(SO4)2, HgSO4, Ce(SO4)2 and Pb(SO4)2. Chemical communications (Cambridge, England).
DOI: 10.1039/b500147a
|




























































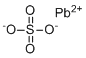


 GHS07;GHS08;GHS09 Danger Details
GHS07;GHS08;GHS09 Danger Details