2,4-Pyrimidinediamine 3-oxide, also known as dapsone, was first synthesized and identified by German chemists in the late 19th century. It is derived from the oxidation of dapsone, a synthetic compound that was originally developed as an antimicrobial agent in the early 20th century.
2,4-Pyrimidinediamine 3-oxide exhibits similar pharmacological properties to dapsone. It has shown promise as an antimicrobial agent for the treatment of various bacterial infections, including leprosy, dermatitis herpetiformis, and acne vulgaris. Its mechanism of action involves inhibition of bacterial folate synthesis, which disrupts nucleic acid metabolism and cell proliferation. Dapsone has been incorporated into topical preparations such as creams, gels, and lotions for the treatment of inflammatory skin diseases such as acne and dermatitis. Dapsone has been investigated for use in photodynamic therapy (PDT) to treat cancer and other proliferative diseases. In PDT, a photosensitizer such as dapsone is activated by light of a specific wavelength to generate reactive oxygen species that selectively target and destroy cancer cells while sparing healthy tissue. Free radicals and reactive oxygen species can cause oxidative stress and cell damage. Dapsone exhibits antioxidant activity by scavenging free radicals and reactive oxygen species, helping to protect cells and tissues from oxidative damage caused by environmental toxins, UV radiation, and inflammatory processes, thereby reducing the risk of chronic diseases and aging-related disorders. Dapsone oxide has been used as a diagnostic reagent in biochemical assays and immunoassays to detect and quantify specific analytes in biological samples. Its stable chemical properties and high specificity make it suitable for a variety of analytical techniques, including enzyme-linked immunosorbent assays (ELISA) and chromatography.
References
2023. A Full Factorial Design to Optimize Aminexil Nano Lipid Formulation to Improve Skin Permeation and Efficacy Against Alopecia. AAPS PharmSciTech, 24(1).
DOI: 10.1208/s12249-023-02500-3
2020. Chromatographic Methods for the Determination of Aminexil, Pyridoxine, and Niacinamide in a Novel Cosmetic Hair Preparation. Journal of AOAC INTERNATIONAL, 103(4).
DOI: 10.1093/jaoacint/qsaa013
2015. Stability indicating RP-HPLC method development and validation for the simultaneous determination of aminexil and minoxidil in pharmaceutical dosage form. Annales Pharmaceutiques Françaises, 73(2).
DOI: 10.1016/j.pharma.2014.10.005
|

































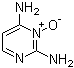
 GHS07 Warning Details
GHS07 Warning Details