Online Database of Chemicals from Around the World
| Nanjing Taiye Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taiyechem.com | |||
 | +86 (25) 5233-7972 | |||
 | +86 (25) 5233-7971 | |||
 | market@taiyechem.com taiyechem@gmail.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Earlier HK Company Limited | China | |||
|---|---|---|---|---|
 | www.earlierchem.com | |||
 | +86 (10) 6142-6769 +86 18616907430 | |||
 | +86 (10) 6142-6769 | |||
 | info@earlierchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| DQ Pure | Indonesia | |||
|---|---|---|---|---|
 | dqpure.com | |||
 | +86 13588024337 | |||
 | lu@dqpure.com | |||
 | WhatsApp:+8613588024337 | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2026 | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Xinyu Dongpeng Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rubidium-cs.com | |||
 | +86 (790) 686-4666 646-4661 646-4662 | |||
 | +86 (790) 646-4663 | |||
 | jxxydpc@163.com | |||
| Chemical manufacturer since 2000 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Rubidium sulfate |
| Molecular Structure | 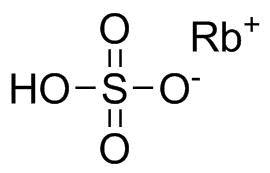 |
| Molecular Formula | Rb2SO4 |
| Molecular Weight | 267.00 |
| CAS Registry Number | 7488-54-2 |
| EC Number | 231-301-7 |
| SMILES | OS(=O)(=O)[O-].[Rb+] |
| Density | 3.613 g/mL (Expl.) |
|---|---|
| Melting point | 1050 $degree$C (Expl.) |
| Boiling point | 1700 $degree$C (Expl.) |
| Solubility | water 48.2% w/w in 20 $degree$C, slightly soluble in NH3, insoluble in acetone (Expl.) |
| Refraction index | 1.513 (Expl.) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P280-P264-P305+P351+P338-P321-P332+P313-P337+P313 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Rubidium sulfate is an inorganic salt with the chemical formula Rb2SO4. It consists of rubidium cations (Rb+) and sulfate anions (SO42−). As an alkali metal sulfate, it is structurally and chemically similar to potassium sulfate and sodium sulfate, but it is less commonly encountered due to the relatively low natural abundance and higher cost of rubidium. The sulfate ion in rubidium sulfate has a tetrahedral geometry, with a central sulfur atom covalently bonded to four oxygen atoms. The negative charge is delocalized over the oxygen atoms through resonance, which contributes to the stability of the anion. In the solid state, rubidium ions and sulfate ions form an ionic crystal lattice held together by electrostatic attraction between Rb+ and SO42−. Rubidium sulfate can be prepared by neutralizing rubidium hydroxide or rubidium carbonate with sulfuric acid. In these acid–base reactions, the strong base (RbOH or Rb2CO3) reacts with sulfuric acid to form rubidium sulfate and water, with carbon dioxide released in the case of carbonate. The resulting solution can be evaporated to yield crystalline rubidium sulfate. In aqueous solution, rubidium sulfate dissociates completely into rubidium and sulfate ions. The solution is typically neutral because both ions are derived from a strong base (RbOH) and a strong acid (H2SO4), resulting in negligible hydrolysis. This behavior is characteristic of most alkali metal sulfates. Rubidium sulfate is primarily used in laboratory and research settings rather than large-scale industrial applications. It serves as a source of rubidium ions for chemical studies and is used in investigations involving alkali metal chemistry, crystallography, and spectroscopy. Rubidium compounds are also of interest in atomic physics and quantum research, where precise control of alkali metal species is required. In materials science, rubidium sulfate has been studied in the context of crystal growth and ionic conductivity. Like other alkali metal sulfates, it can form well-defined crystalline structures that are useful for studying ionic interactions and lattice behavior. However, practical applications are limited compared with more common sulfate salts such as potassium sulfate. From a physicochemical perspective, rubidium sulfate reflects trends in the alkali metal series. As the ionic radius increases from sodium to rubidium, lattice energies decrease, which influences solubility and crystal stability. Rubidium sulfate is moderately soluble in water, though less so than sodium sulfate. Its high molar mass and large cation size also affect its thermal and structural properties. Historically, rubidium compounds were first identified in the mid-19th century using flame spectroscopy techniques developed by Robert Bunsen and Gustav Kirchhoff. While initial studies focused on elemental identification, later research expanded into the chemistry of rubidium salts, including sulfates, halides, and carbonates, to understand periodic trends among alkali metals. Overall, rubidium sulfate is a simple inorganic ionic compound composed of rubidium and sulfate ions. It is primarily used in research contexts as a source of rubidium and as a model compound for studying alkali metal sulfate chemistry, crystal structures, and ionic behavior in solid-state and solution systems. References 2014. Growth and various characterizations of LiHSO4 single crystals. Journal of Materials Science: Materials in Electronics. DOI: 10.1007/s10854-014-2561-3 2007. Origin of dielectric relaxations in KHSeO4 above room temperature. Ionics. DOI: 10.1007/s11581-013-0994-y 2005. 40A-2 RbHSO4 [F]. Inorganic Substances other than Oxides. DOI: 10.1007/10552342_37 |
| Market Analysis Reports |