Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shandong Jiachen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiachenchem.net | |||
 | +86 19854180086 | |||
 | 476157612@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Weiyel Inc | China | |||
|---|---|---|---|---|
 | www.weiyel.com | |||
 | +44 7421898692 | |||
 | ivy@weiyelbncc.com | |||
 | QQ Chat | |||
 | WeChat: +8615672728568 | |||
 | WhatsApp:+44 7421898692 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Cyanopyridine |
|---|---|
| Name | 1,1-Dichloroethylene |
| Synonyms | Vinylidene chloride |
| Molecular Structure | 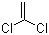 |
| Molecular Formula | C2H2Cl2 |
| Molecular Weight | 96.94 |
| CAS Registry Number | 75-35-4 |
| EC Number | 200-864-0 |
| SMILES | C=C(Cl)Cl |
| Solubility | 0.04 g/100g |
|---|---|
| Density | 1.213 g/mL |
| Melting point | -122 °C |
| Boiling point | 30-32 °C |
| Flash point | -25 °C |
| Hazard Symbols |    GHS02;GHS08;GHS07 Danger Details GHS02;GHS08;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H224-H351-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P303+P361+P353-P304+P340-P305+P351+P338-P317-P318-P319-P330-P337+P317-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1303 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1,1-Dichloroethylene, also known as vinylidene chloride, is a chemical compound with the formula C2H2Cl2. This colorless, flammable liquid has a sweet odor and is classified as a chlorinated hydrocarbon. It was first synthesized in the early 19th century, with the discovery attributed to the work of chemists investigating the properties of chlorinated alkenes. Its commercial production began in the mid-20th century, primarily due to the increasing demand for versatile and effective industrial solvents. 1,1-Dichloroethylene has found numerous applications across various industries. One of its primary uses is as a solvent in the formulation of adhesives, coatings, and paint removers. Its ability to dissolve a wide range of organic materials makes it valuable in these formulations, where it enhances the effectiveness of the products. In addition to serving as a solvent, 1,1-dichloroethylene is utilized in the production of fluoropolymers and other chemicals, functioning as an intermediate in the synthesis of more complex compounds. Another significant application of 1,1-dichloroethylene lies in the field of plastics manufacturing. It is employed in the production of polyvinylidene chloride (PVDC), a polymer known for its excellent barrier properties against moisture, oxygen, and other gases. This characteristic makes PVDC widely used in food packaging and preservation, providing an effective means to extend the shelf life of perishable goods. The use of 1,1-dichloroethylene in creating PVDC has revolutionized packaging technologies, enhancing food safety and reducing waste. In addition to industrial applications, 1,1-dichloroethylene has also been utilized in laboratory settings as a reagent in organic synthesis. Its reactivity allows for various chemical transformations, including polymerization and cross-linking reactions, contributing to the development of new materials and compounds. Furthermore, it has been investigated for its potential applications in the synthesis of agrochemicals and pharmaceuticals. Despite its usefulness, 1,1-dichloroethylene is subject to regulatory scrutiny due to its environmental and health impacts. It is classified as a hazardous air pollutant by the United States Environmental Protection Agency (EPA) and is associated with potential risks, including carcinogenicity and reproductive toxicity. Consequently, industries utilizing this compound must adhere to strict guidelines and regulations to mitigate its release into the environment and minimize exposure to workers. Efforts to find safer alternatives to 1,1-dichloroethylene have led to research into green chemistry solutions and the development of less toxic solvents. As industries evolve and seek to reduce their environmental footprint, the demand for safer and more sustainable alternatives continues to grow. In summary, 1,1-dichloroethylene is a significant chemical compound with a rich history of discovery and diverse applications in various industrial and research contexts. While it has played a vital role in the development of new materials and products, ongoing concerns regarding its environmental and health impacts underscore the need for responsible use and the exploration of safer alternatives. References 1984. 1,1-Dichloroethylene: An Apoptotic Hepatotoxin?. Environmental Health Perspectives, 57. DOI: 10.1289/ehp.8457313 2017. Cometabolic degradation of dichloroethenes by Comamonas testosteroni RF2. Chemosphere, 187. DOI: 10.1016/j.chemosphere.2017.07.156 2009. Large-scale production of bacterial consortia for remediation of chlorinated solvent-contaminated groundwater. Journal of Industrial Microbiology & Biotechnology, 36(9). DOI: 10.1007/s10295-009-0600-5 |
| Market Analysis Reports |