Online Database of Chemicals from Around the World
| Hunan Astar Bio-chemicals Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.astarchem.com | |||
 | +86 (10) 5831-9740 | |||
 | +86 (10) 5831-9810 | |||
 | export02@astarchem.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hubei Research Institute of Chemistry | China | |||
|---|---|---|---|---|
 | www.haisopharm.com | |||
 | +86 (27) 8742-2225 | |||
 | +86 (27) 8749-6702 | |||
 | info@haisopharm.com | |||
| Chemical manufacturer since 1973 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Henan Topfond Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.topfond.com | |||
 | +86 (396) 382-3635 | |||
 | +86 (396) 381-5761 | |||
 | tfyy@meheco.gt.cn export@topfond.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Molcan Corporation | USA | |||
|---|---|---|---|---|
 | www.molcan.com | |||
 | +1 (905) 731-5537 | |||
 | +1 (905) 248-3843 | |||
 | info@molcan.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Apexbio Technology LLC | USA | |||
|---|---|---|---|---|
 | www.apexbt.com | |||
 | +1 (832) 696-8203 | |||
 | +1 (855) 527-3928 | |||
 | info@apexbt.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Depont Molecular Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dpmolecular.com | |||
 | +86 (25) 8421-5993 | |||
 | +86 (25) 8421-7866 | |||
 | jupiter@depontech.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Zhejiang Sanhe Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjshsw.com | |||
 | +86 (570) 496-5558 | |||
 | +86 (570) 496-9333 | |||
 | wy@zjshsw.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Nanjing Xize Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njxizebio.com | |||
 | +86 (25) 6602-3220 | |||
 | +86 (25) 6602-3220 | |||
 | sale@njxizebio.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Hubei Marvel-bio Medicine Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.marvel-bio.com | |||
 | +86 (027) 8773-8507 | |||
 | maoerwo123@163.com | |||
 | QQ Chat | |||
 | WeChat: kangbao813 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Zhejiang Jiangbei Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiangbei.com | |||
 | +86 (415) 8878-6567 | |||
 | +86 (415) 8878-6538 | |||
 | trade@jiangbei.com | |||
| Chemical manufacturer since 1991 | ||||
| CNH Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.cnhtechnologies.com | |||
 | +1 (781) 933-0362 | |||
 | +1 (781) 933-1839 | |||
 | info@cnhtechnologies.com | |||
| Chemical manufacturer | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Yung Zip Chemical Co., Ltd. | Taiwan | |||
|---|---|---|---|---|
 | www.yungzip.com | |||
 | +886 (4) 2681-8780 2681-1674 | |||
 | +886 (4) 2681-2911 | |||
 | yspgyzc@yungshingroup.com | |||
| Chemical manufacturer since 1978 | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| A.S. Chemical Laboratories Inc. | Canada | |||
|---|---|---|---|---|
 | www.aschemicals.com | |||
 | +1 (905) 669-7472 | |||
 | sales@aschemicals.com | |||
| Chemical manufacturer since 1997 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Circulatory system medication >> Regulating blood lipids |
|---|---|
| Name | Lovastatin |
| Synonyms | 1,2,3,7,8,8a-Hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl]-1-naphthalenyl 2-methylbutanoate |
| Molecular Structure | 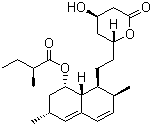 |
| Molecular Formula | C24H36O5 |
| Molecular Weight | 404.54 |
| CAS Registry Number | 75330-75-5 |
| EC Number | 692-955-0 |
| SMILES | CC[C@H](C)C(=O)O[C@H]1C[C@H](C=C2[C@H]1[C@H]([C@H](C=C2)C)CC[C@@H]3C[C@H](CC(=O)O3)O)C |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 175 °C (Expl.) |
| Boiling point | 559.2±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 185.3±23.6 °C (Calc.)* |
| Solubility | DMSO 8 mg/mL, Water $lessThan$1 mg/mL (Expl.) |
| Index of refraction | 1.532 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H351-H361-H372 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P270-P272-P280-P301+P317-P302+P352-P318-P319-P321-P330-P333+P317-P362+P364-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Lovastatin is a naturally occurring statin, classified as a member of the fungal polyketide family, used clinically as a cholesterol-lowering agent. Its molecular formula is C24H36O5. Structurally, lovastatin contains a hexahydro-naphthalene core fused to a lactone ring and a side chain that mimics the substrate of HMG-CoA reductase, the key enzyme in cholesterol biosynthesis. It appears as a white to off-white crystalline powder that is practically insoluble in water but soluble in organic solvents such as ethanol, methanol, and dichloromethane. The discovery of lovastatin dates to the 1970s when it was isolated from the fungus *Aspergillus terreus*. Its development was driven by the need for pharmacological agents to lower plasma cholesterol and reduce the risk of cardiovascular disease. Lovastatin functions as a competitive inhibitor of HMG-CoA reductase, decreasing the conversion of HMG-CoA to mevalonate, an early and rate-limiting step in cholesterol biosynthesis. This results in lower levels of low-density lipoprotein cholesterol (LDL-C) and modest increases in high-density lipoprotein cholesterol (HDL-C). The industrial production of lovastatin is primarily achieved through fermentation using *Aspergillus* species under controlled conditions. The fungus is cultured in nutrient-rich media to optimize polyketide synthesis, and the lovastatin is subsequently extracted using organic solvents. Purification involves crystallization or chromatographic techniques to yield the biologically active lactone form. Semi-synthetic derivatives, such as simvastatin, are prepared by chemical modification of lovastatin to enhance pharmacokinetic or pharmacodynamic properties. Chemically, lovastatin exists as a lactone that can be hydrolyzed in vivo to the corresponding β-hydroxy acid, which is the active inhibitor of HMG-CoA reductase. The molecule contains multiple chiral centers, and its biological activity is highly stereospecific. The lactone ring and hexahydro-naphthalene scaffold allow binding to the enzyme’s active site, mimicking the natural substrate. Lovastatin is stable under normal storage conditions but can undergo hydrolysis or oxidation under strong acidic, basic, or oxidative conditions. In practical applications, lovastatin is prescribed to patients with hypercholesterolemia and dyslipidemia to reduce cardiovascular risk. It is administered orally and metabolized primarily by hepatic cytochrome P450 enzymes. Beyond its cholesterol-lowering effects, lovastatin has been studied for potential pleiotropic benefits, including anti-inflammatory, antioxidant, and endothelial function-improving activities. Lovastatin is also used as a lead compound in medicinal chemistry for the development of other statins with improved potency, bioavailability, or reduced side effects. Physically, lovastatin should be stored in a cool, dry environment, protected from light and moisture to prevent hydrolysis of the lactone ring. Handling precautions include avoiding inhalation or contact with skin and eyes, as with other pharmaceutical powders. The compound is non-volatile and stable under ambient laboratory conditions. Overall, lovastatin is a clinically significant natural statin that serves as a model for cholesterol-lowering therapy. Its ability to inhibit HMG-CoA reductase, combined with the polyketide structure derived from fungal biosynthesis, makes it a cornerstone in cardiovascular pharmacotherapy and a template for the development of additional statin drugs. References 2025. Lovastatin inhibits adipocyte-associated increased proliferation, EMT and aggressiveness of breast cancer cells. Medical Oncology, 42(8). DOI: 10.1007/s12032-025-02874-3 2025. Heat capacity and thermodynamic functions of crystalline and amorphous forms of Lovastatin. Scientific Reports. DOI: 10.1038/s41598-025-05075-0 |
| Market Analysis Reports |