Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Jiyuan Ruiyang Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | en.jyrysy.com | |||
 | +86 (391) 666-0065 | |||
 | +86 (391) 666-0065 | |||
 | ruiyangaaron@163.com | |||
 | WeChat: +8615239782640 | |||
 | WhatsApp:+8615239782640 | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Boulder Scientific Company | USA | |||
|---|---|---|---|---|
 | www.bouldersci.com | |||
 | +1 (970) 535-4494 | |||
 | +1 (970) 535-4584 | |||
 | sales@bouldersci.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal chlorides and salts |
|---|---|
| Name | Titanium tetrachloride |
| Synonyms | Tetrachlorotitanium; Titanic chloride |
| Molecular Structure | 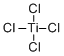 |
| Molecular Formula | TiCl4 |
| Molecular Weight | 189.68 |
| CAS Registry Number | 7550-45-0 |
| EC Number | 231-441-9 |
| SMILES | Cl[Ti](Cl)(Cl)Cl |
| Density | 1.726 |
|---|---|
| Melting point | -25 °C |
| Boiling point | 135-136 °C |
| Flash point | 8 °C |
| Water solubility | reacts |
| Hazard Symbols |     GHS05;GHS06;GHS07;GHS08 Danger Details GHS05;GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H317-H318-H330-H335-H370-H372 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P272-P280-P284-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P308+P316-P316-P317-P319-P320-P321-P333+P317-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1838; UN 2443 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Titanium tetrachloride (TiCl₄) was first synthesized in 1825 by Swedish chemist Jöns Jacob Berzelius. Berzelius, a pioneer in the field of chemical elements, discovered TiCl₄ while studying titanium compounds. This discovery was crucial to the understanding of titanium chemistry and laid the foundation for its widespread industrial applications. Titanium tetrachloride is an important compound with the following properties: it appears as a colorless liquid, has a boiling point of 136.4°C (277.5°F), a melting point of -24.5°C (-11.1°F), a density of 1.62 g/cm³, and reacts with water to form titanium dioxide (TiO₂) and hydrochloric acid (HCl). TiCl₄ is very reactive, especially with water, forming a white titanium dioxide precipitate and releasing hydrochloric acid. Its reactivity makes it a valuable compound in a variety of chemical processes. The main use of titanium tetrachloride is in the production of titanium dioxide. It is a precursor for the production of titanium dioxide (TiO₂), a white pigment widely used in paints, coatings, and plastics. The production process involves chlorinating titanium ore to obtain TiCl₄, which is then oxidized to form TiO₂. This pigment is valued for its brightness and opacity. TiCl₄ is used as a catalyst in various chemical reactions. It is used as a catalyst in the Ziegler-Natta polymerization process to produce polymers such as polypropylene and polyethylene. This catalytic application is essential for the production of high-performance plastics used in numerous industrial and consumer products. In the metallurgical industry, TiCl₄ is a precursor for the production of titanium metal via the Kroll process. This process reduces TiCl₄ with magnesium to obtain titanium metal, which is essential for aerospace, automotive, and medical applications due to its strength and corrosion resistance. TiCl₄ is used as a reagent in organic chemistry to synthesize a variety of compounds. The ability of TiCl₄ to form coordination compounds makes it valuable in chemical synthesis and catalysis. TiCl₄ is used as a flocculant in water treatment processes to help remove impurities and particles from water. Due to its reactive nature, handling titanium tetrachloride requires careful safety measures, wearing appropriate personal protective equipment including gloves, goggles and lab coats, and avoiding contact with skin and eyes. Work in a well-ventilated area or use a fume hood to prevent inhalation of fumes. Store in a sealed container in a cool, dry place away from moisture and incompatible materials. Proper storage prevents unintended reactions and maintains compound stability. Environmental and health effects must be considered when handling TiCl₄, and spills and leaks should be carefully managed to prevent environmental contamination. The reaction of TiCl₄ with water results in the release of hydrochloric acid, which needs to be controlled to avoid environmental damage. Exposure to titanium tetrachloride may cause irritation to the respiratory system, skin, and eyes. Following safety procedures is essential to minimize health risks. References 1986. Inhalation toxicity study on rats exposed to titanium tetrachloride atmospheric hydrolysis products for two years. Toxicology and Applied Pharmacology, 83(1). DOI: 10.1016/0041-008x(86)90320-0 2012. Anionic polymer compound bioflocculant as a coagulant aid with aluminum sulfate and titanium tetrachloride. Bioresource Technology, 108. DOI: 10.1016/j.biortech.2012.01.012 2017. Electrodeposition of SnO2 on FTO and its Application in Planar Heterjunction Perovskite Solar Cells as an Electron Transport Layer. Nanoscale Research Letters, 12(1). DOI: 10.1186/s11671-017-2247-x |
| Market Analysis Reports |