Online Database of Chemicals from Around the World
| Shandong Hongkun Group | China | |||
|---|---|---|---|---|
 | www.hongkungroup.com | |||
 | +86 (718) 395-3311 | |||
 | +86 (718) 461-7902 | |||
 | mwu59@yahoo.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Panjin Jiahe Shengshi Pharmaceutical Technology Co., Ltd | China | |||
|---|---|---|---|---|
 | www.jhssw.com | |||
 | +86 13032431717 | |||
 | sale@jhssw.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Wuhan Yuancheng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuanchengtechnology.com | |||
 | +86 17282536078 | |||
 | +86 17282536078 | |||
 | 3042184429@qq.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17282536078 | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Autech Industry Co., Limited | China | |||
|---|---|---|---|---|
 | www.autechindustry.com | |||
 | +86 (151) 6531-0911 | |||
 | sales@autechindustry.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Spectrum Info Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.spec-info.com | |||
 | +38 (044) 573-5462 | |||
 | +38 (044) 573-2397 | |||
 | spec-info@spec-info.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Classification | API >> Digestive system medication >> Laxative antidiarrheal |
|---|---|
| Name | Phenolphthalein |
| Synonyms | 2-[Bis(4-hydroxyphenyl)methyl]benzoic acid; 3,3-Bis(4-hydroxyphenyl)-1(3H)-isobenzofuranone; 3,3-Bis(4-hydroxyphenyl)phthalide |
| Molecular Structure | 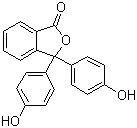 |
| Molecular Formula | C20H14O4 |
| Molecular Weight | 318.33 |
| CAS Registry Number | 77-09-8 |
| EC Number | 201-004-7 |
| SMILES | C1=CC=C2C(=C1)C(=O)OC2(C3=CC=C(C=C3)O)C4=CC=C(C=C4)O |
| Density | 1.299 |
|---|---|
| Melting point | 258-263 °C |
| Water solubility | <0.1 g/100 mL |
| Hazard Symbols |  GHS08 Danger Details GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H350-H341-H361f Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P280-P318-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Phenolphthalein, with the chemical formula C20H14O4, is a widely recognized and versatile compound in chemistry, known for its use as a pH indicator and in various other applications. It was first synthesized in 1871 by the German chemist Rudolf Vergin. The compound is derived from the reaction of phenol with phthalic anhydride under acidic conditions, leading to the formation of a colorless crystalline solid that becomes pink to red in basic solutions. Phenolphthalein is best known for its role as an acid-base indicator in titrations. It changes color in the pH range of approximately 8.2 to 10.0. In acidic solutions, phenolphthalein remains colorless, but as the pH increases and the solution becomes more basic, the compound turns pink. This color change is attributed to the ionization of phenolphthalein in the basic environment, where it forms a colored anionic species. This property makes it a valuable tool in analytical chemistry for determining the endpoint of titrations involving weak acids and strong bases. In addition to its use in titrations, phenolphthalein has applications in various fields, including medicine and industry. Historically, it was used as a laxative due to its ability to stimulate bowel movements. However, its use in this capacity has diminished due to concerns over safety and potential side effects. In modern times, phenolphthalein is primarily used in laboratory settings and in the manufacturing of certain dyes and chemicals. Phenolphthalein's role extends to educational demonstrations and chemical experiments. It is commonly used in classroom settings to illustrate the concept of pH indicators and to visually demonstrate the changes that occur during acid-base reactions. Its vivid color change makes it an effective teaching tool for visualizing the principles of acid-base chemistry. In industrial applications, phenolphthalein is used as a reagent in the synthesis of other chemicals and as a component in certain types of pH-sensitive materials. Its stability and color-changing properties make it useful in various chemical formulations and quality control processes. Overall, phenolphthalein remains a significant compound in both academic and practical chemistry. Its ability to indicate pH changes makes it an invaluable tool in titrations and educational demonstrations, while its applications in industry and historical use as a laxative highlight its versatility. References 1984. Kinetic study of beta-cyclodextrin-dye system by high-pressure temperature-jump method. The Journal of Biochemistry, 96(1). DOI: 10.1093/oxfordjournals.jbchem.a134830 2011. Laxative-Induced Buttock Dermatitis. The Journal of Emergency Medicine, 40(2). DOI: 10.1016/j.jemermed.2008.06.013 1964. The diagnostic value of the twenty-four hour pH gastric analysis—A preliminary report. Irish Journal of Medical Science (1926-1967), 39(10). DOI: 10.1007/bf02944846 |
| Market Analysis Reports |