Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic scandium, tantalum, thallium, tungsten, antimony, lanthanum, lead, vanadium, molybdenum, chromium, ytterbium, etc. |
|---|---|
| Name | Tantalum chloride |
| Synonyms | Pentachlorotantalum; Tantalum chloride; Tantalum pentachloride |
| Molecular Structure | 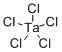 |
| Molecular Formula | TaCl5 |
| Molecular Weight | 358.21 |
| CAS Registry Number | 7721-01-9 |
| EC Number | 231-755-6 |
| SMILES | Cl[Ta](Cl)(Cl)(Cl)Cl |
| Solubility | Soluble (absolute alcohol and KOH) |
|---|---|
| Density | 3.68 g/mL |
| Melting point | 214-216 °C |
| Boiling point | 242 °C |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P260-P271-P280-P301+P312-P303+P361+P353-P305+P351+P338 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Tantalum chloride (TaCl₅) is an important compound in the fields of materials science and catalysis. It was discovered in the early 20th century and has become key to a variety of industrial and technological applications due to its unique properties. Tantalum chloride was first synthesized in 1922 by German chemist Werner Fischer. Fischer's work involved chlorinating tantalum (a rare metal named after Tantalus from Greek mythology) to produce tantalum chloride. The compound is a white or yellowish powder and has since been studied for its unique chemical behavior and usefulness. Tantalum chloride is known for its high melting point (about 2850°C) and is stable at high temperatures. It is highly reactive with water, forming tantalum pentoxide (Ta₂O₅) and hydrochloric acid. This property is fully utilized in various chemical processes. Tantalum chloride is also soluble in alcohols and certain organic solvents, which facilitates its use in various chemical reactions. Tantalum chloride is a valuable catalyst in organic synthesis. It is used in the production of various chemicals, including petrochemicals and pharmaceuticals. Its ability to act as a Lewis acid enables it to effectively promote reactions such as alkylation and acylation. In materials science, tantalum chloride is a precursor for the deposition of tantalum metal and tantalum-based compounds. These materials have excellent electrical conductivity and high melting points, which are necessary for the production of high-performance electronic products, including capacitors and semiconductors. Tantalum chloride plays an important role in the electronics industry, especially in the production of tantalum capacitors. These capacitors are stable and reliable in high-temperature environments , ideal for use in advanced electronics and aerospace applications. The compound is also used to produce tantalum-based coatings and alloys. These materials are highly resistant to corrosion and wear, making them ideal for use in harsh environments, such as those encountered in chemical processing and the aerospace industry. Ongoing research is exploring new applications for tantalum chloride, particularly in the areas of nanotechnology and advanced materials. Innovations in processing and application techniques are expected to expand its role in emerging technologies and sustainable practices. In summary, tantalum chloride is a versatile compound with important applications in catalysis, materials science, electronics, and coatings. Its discovery paved the way for major advances in various industrial fields, and its future applications are expected to continue to drive technological advancements. References 2023. TaCl5 in the synthesis of amides from saturated monobasic carboxylic acids and functionally substituted primary aromatic amines. Russian Chemical Bulletin, 72(10). DOI: 10.1007/s11172-023-4032-6 1956. Die Reduktion der Pentachloride und Pentabromide des Niobs und Tantals mit Wasserstoff in der elektrischen Entladung. Monatshefte für Chemie und verwandte Teile anderer Wissenschaften, 87(5). DOI: 10.1007/bf00899589 1954. Die halbquantitative Wolframbestimmung in Niob-Tantal-Präparaten und die Abtrennung des Wolframs von Niob und Tantal. Fresenius' Zeitschrift für analytische Chemie, 141(1). DOI: 10.1007/bf00433663 |
| Market Analysis Reports |