Online Database of Chemicals from Around the World
| Xi'an Yinherb Bio-tech Co., Ltd | China | |||
|---|---|---|---|---|
 | yinherb.ecer.com | |||
 | +86 (29) 6333-2330 | |||
 | sales@yinherb-nutraceuticals.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shandong Zhishang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhishangchemical.com | |||
 | +86 17653113209 13065062079 | |||
 | sales001@sdzschem.com sales002@sdzschem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shandong Look Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sdlookchem.com | |||
 | +86 17653113219 | |||
 | sales01@sdlookchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Classification | Organic raw materials >> Organic derivative of hydrazine or hydrazine |
|---|---|
| Name | 2-Oxo-4-phenyl-1-pyrrolidineacetic acid hydrazide |
| Synonyms | 2-(2-oxo-4-phenylpyrrolidin-1-yl)acetohydrazide |
| Molecular Structure | 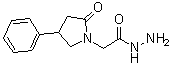 |
| Molecular Formula | C12H15N3O2 |
| Molecular Weight | 233.27 |
| CAS Registry Number | 77472-71-0 |
| SMILES | C1C(CN(C1=O)CC(=O)NN)C2=CC=CC=C2 |
| Solubility | Very slightly soluble (0.51 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.243±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 154-155 °C** |
| Boiling point | 529.4±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 274.0±30.1 °C (Calc.)* |
| Index of refraction | 1.587 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2018 ACD/Labs) |
| ** | Glozman, O. M. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P312+P330 Details |
| SDS | Available |
|
2-Oxo-4-phenyl-1-pyrrolidineacetic acid hydrazide is a chemical compound belonging to the class of hydrazides, characterized by the presence of a hydrazide functional group attached to a pyrrolidine ring. The structure includes a pyrrolidine ring substituted at the 4-position with a phenyl group and bearing a keto group (=O) at the 2-position. The acetic acid moiety is linked through the nitrogen atom of the pyrrolidine, and this acetic acid is further functionalized as a hydrazide. This compound has significance in medicinal chemistry due to its potential biological activities. The pyrrolidine ring is a common motif in many bioactive molecules, contributing to the compound’s ability to interact with various biological targets, such as enzymes or receptors. The hydrazide group imparts chemical reactivity that can be exploited in drug design, including the formation of hydrazones or other derivatives. In research, 2-oxo-4-phenyl-1-pyrrolidineacetic acid hydrazide has been investigated for its possible use as a precursor in the synthesis of more complex molecules with pharmacological relevance. Its hydrazide functionality allows it to be employed in condensation reactions with aldehydes or ketones to form hydrazones, which may exhibit various biological activities including antimicrobial, antitubercular, or anticancer properties. Synthetically, the compound is typically prepared by the reaction of the corresponding acid chloride or ester of 2-oxo-4-phenyl-1-pyrrolidineacetic acid with hydrazine or hydrazine derivatives. This method provides a straightforward pathway to the hydrazide, allowing for subsequent functionalization or biological evaluation. The compound’s physicochemical properties, such as solubility and stability, make it suitable for use in various chemical and pharmaceutical applications. Its reactivity profile enables further chemical modifications, making it a versatile intermediate in drug development pipelines. Overall, 2-oxo-4-phenyl-1-pyrrolidineacetic acid hydrazide represents an important chemical scaffold with potential utility in medicinal chemistry, particularly as a building block for synthesizing molecules with diverse biological activities. References 2023. Radiolabeling and evaluation of fonturacetam hydrazide as a radiotracer for visualization of brain function. Journal of Radioanalytical and Nuclear Chemistry, 332(6). DOI: 10.1007/s10967-023-08966-6 |
| Market Analysis Reports |