Potassium nitrate, commonly known by its chemical formula KNO₃, is a naturally occurring mineral and an important chemical with applications spanning agriculture, medicine, food preservation, and pyrotechnics. Its discovery dates back thousands of years, as it naturally forms in arid environments and can accumulate in the soil as a result of bacterial activity on decomposing organic matter. Historically, potassium nitrate was a key component of saltpeter, an important material for gunpowder production. Ancient civilizations, particularly in China and the Middle East, valued potassium nitrate as a critical substance for both military and agricultural purposes.
In agriculture, potassium nitrate is prized as a highly effective fertilizer. It provides two essential nutrients: potassium and nitrogen. Potassium is crucial for plant health, aiding in the regulation of water uptake, enzyme activation, and photosynthesis, while nitrogen is a primary component of amino acids and chlorophyll, supporting plant growth and development. Potassium nitrate’s water solubility makes it an ideal fertilizer, as it can be delivered to plants through irrigation systems, ensuring that the nutrients are readily available in the soil. It is especially beneficial for crops like fruits and vegetables, which have high potassium demands and thrive with a balanced nitrogen supply.
In medicine, potassium nitrate has been historically used for its diuretic and anti-inflammatory properties. While its medicinal use is less common today, it still finds a role in dental health, particularly in toothpaste formulated for sensitive teeth. Potassium ions in potassium nitrate help to block the transmission of pain signals from the tooth nerves, reducing discomfort caused by sensitive teeth. This application has made potassium nitrate a popular ingredient in many over-the-counter dental products.
Potassium nitrate also serves as a preservative and curing agent in the food industry. It has been used for centuries in curing meats such as ham, bacon, and sausages, as it inhibits bacterial growth, particularly that of Clostridium botulinum, which can cause botulism, a severe form of food poisoning. In addition to preserving color and flavor in cured meats, potassium nitrate helps ensure food safety during extended storage. However, due to health concerns, its use in food is now regulated, and many producers have reduced its usage in favor of other preservatives.
One of the most prominent historical and modern applications of potassium nitrate is in pyrotechnics and explosives. When combined with sulfur and charcoal, potassium nitrate forms gunpowder, a mixture that has been used in firearms, fireworks, and construction blasting for centuries. As an oxidizing agent, potassium nitrate supports combustion by releasing oxygen, making it a critical component in pyrotechnic formulations. In fireworks, it contributes to the production of colorful displays, while in construction, it is used in controlled blasting. Potassium nitrate’s role in gunpowder production shaped many historical developments, especially in warfare, and continues to be significant in various industries today.
Modern industrial uses of potassium nitrate extend to glass manufacturing, ceramics, and metal treatment. It acts as a flux in the glass and ceramic industries, where it helps lower the melting point of raw materials, leading to smoother and more uniform products. In metal treatment, potassium nitrate is involved in processes like heat-treating steel, where it contributes to hardening the metal surface, enhancing durability and resistance to wear. These applications showcase the versatility of potassium nitrate across various sectors.
Despite its benefits, potassium nitrate must be handled with care. It is a strong oxidizer and, when improperly stored, can be hazardous, particularly in settings where there is risk of accidental combustion. In agriculture, overuse of potassium nitrate as a fertilizer can lead to soil and water pollution, contributing to nutrient imbalances and environmental degradation. As such, strict guidelines and handling practices are essential to ensure safe and sustainable use of potassium nitrate in all its applications.
Potassium nitrate’s long history and diverse uses highlight its importance as a versatile and impactful chemical. Whether enhancing agricultural productivity, preserving food, treating sensitive teeth, or producing fireworks, potassium nitrate remains a critical resource in both traditional and modern industries.
References
1984. Chemical composition for saltpeter. Journal of the American Academy of Dermatology.
DOI: 10.1016/s0190-9622(84)80529-0
2003. Evaluation of the efficacy of two potassium nitrate bioadhesive gels (5% and 10%) in the treatment of dentine hypersensitivity. A randomised clinical trial. Journal of Clinical Periodontology.
DOI: 10.1034/j.1600-051x.2003.20077.x
|








































































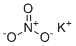

 GHS03;GHS07 Danger Details
GHS03;GHS07 Danger Details