Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shabbir Chemicals | India | |||
|---|---|---|---|---|
 | www.shabbirchemicals.com | |||
 | +91 9441800179 | |||
 | shabbirchemic@gmail.com | |||
 | WhatsApp:+919441800179 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Organometallic salt |
|---|---|
| Name | Sodium amide |
| Molecular Structure | 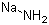 |
| Molecular Formula | NaNH2 |
| Molecular Weight | 39.01 |
| CAS Registry Number | 7782-92-5 |
| EC Number | 231-971-0 |
| SMILES | [NH2-].[Na+] |
| Density | 1.37 g/mL (Expl.) |
|---|---|
| Melting point | 210 °C (Expl.) |
| Boiling point | 400 °C (Expl.) |
| Solubility | water: reacts very violently, even explosively (Expl.) |
| Hazard Symbols |    GHS02;GHS05;GHS09 Danger Details GHS02;GHS05;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H260-H261-H314-H318-H400 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P223-P231+P232-P260-P264-P264+P265-P273-P280-P301+P330+P331-P302+P335+P334-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P370+P378-P391-P402+P404-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1425; UN 1390 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Sodium amide is an inorganic compound with the chemical formula NaNH2. It consists of a sodium cation (Na+) and an amide anion (NH2–), forming a white to gray crystalline solid. Sodium amide is highly reactive, especially toward water and protic solvents, and is widely used as a strong base and nucleophile in organic synthesis. The discovery of sodium amide dates back to the early 20th century during studies of alkali metal reactions with ammonia. It is typically prepared by directly reacting metallic sodium with liquid ammonia under controlled conditions. This reaction produces sodium amide along with hydrogen gas, a process that highlighted the unique ability of alkali metals to deprotonate ammonia and form metal amides. In organic chemistry, sodium amide is primarily employed as a strong, non-nucleophilic base. It is used for deprotonation of weakly acidic hydrogen atoms, such as in terminal alkynes, to generate acetylide ions that can undergo subsequent nucleophilic substitution reactions. Sodium amide is also applied in the synthesis of heterocycles, including aziridines and pyrroles, and in elimination reactions to form alkenes from alkyl halides. Its strong basicity and relatively low nucleophilicity make it a versatile reagent for these transformations. Sodium amide is also used in industrial processes, including the production of dyes, pharmaceuticals, and specialty chemicals. In these applications, it serves as a base to promote condensation, cyclization, and elimination reactions. Due to its high reactivity, particularly with water, oxygen, and alcohols, sodium amide must be handled under anhydrous conditions, typically in liquid ammonia or inert solvents such as tetrahydrofuran (THF). Contact with moisture leads to rapid formation of ammonia and sodium hydroxide, which can be hazardous. Proper protective equipment and handling procedures are essential for safe use. Overall, sodium amide is a highly effective strong base in organic synthesis, valued for its ability to generate reactive intermediates and facilitate a wide range of chemical transformations. Its discovery and development underscore the interplay between inorganic and organic chemistry in creating reagents that expand synthetic capabilities. References 2021. Isolation and reactivity of an elusive diazoalkene. Nature Chemistry, 13(4). DOI: 10.1038/s41557-021-00675-5 2012. Olefination Reactions of Phosphorus-Stabilized Carbon Nucleophiles. Stereoselective Alkene Synthesis. DOI: 10.1007/128_2012_314 2017. DBU-catalyzed, aromatization-oriented, regioselective domino synthesis of 2-aminopyrimidines from β-dicarbonyl compounds, DMF-DMA, and cyanamide. Molecular Diversity, 21(4). DOI: 10.1007/s11030-017-9770-7 |
| Market Analysis Reports |