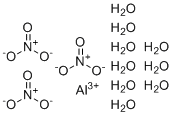
Aluminium nitrate nonahydrate (Al(NO₃)₃·9H₂O) is an inorganic compound commonly used in various industrial and laboratory applications due to its unique chemical properties. This compound is the hydrated form of aluminium nitrate, consisting of aluminium ions (Al³⁺) bound to nitrate ions (NO₃⁻) along with nine water molecules in the crystal structure. Aluminium nitrate nonahydrate is typically produced by dissolving aluminium hydroxide or aluminium oxide in nitric acid, followed by crystallization under controlled conditions to yield the nonahydrate form. This substance was first studied in the late 19th century as part of broader efforts to understand and utilize aluminium compounds in chemical processes.
One of the primary uses of aluminium nitrate nonahydrate is in the production of explosives. It is a key component in the synthesis of ammonium perchlorate, an important oxidizing agent used in solid rocket propellants. In this application, aluminium nitrate nonahydrate provides the necessary aluminium and nitrate ions, which are critical for the creation of energetic materials. It is also used in the preparation of other explosive compounds, where its role as a strong oxidizer contributes to the reactivity and energy output of the mixture.
In addition to its use in explosives, aluminium nitrate nonahydrate is used in the production of other chemicals and materials. It plays a significant role in the manufacture of alumina (Al₂O₃) and other aluminium salts, which are important for a variety of industrial processes. Aluminium nitrate nonahydrate can be used to produce catalysts for chemical reactions and is employed in the synthesis of certain dyes and pigments. Its ability to release aluminium ions upon dissolution makes it valuable in reactions requiring these ions for catalysis or other processes.
The compound is also used in water treatment processes, where it acts as a coagulating agent. Aluminium nitrate nonahydrate helps to remove impurities and suspended particles from water by promoting the aggregation of small particles into larger ones, which can then be removed more easily. This application is particularly useful in the treatment of drinking water and wastewater in municipal and industrial settings, where the removal of contaminants is essential for water purification.
Aluminium nitrate nonahydrate has applications in laboratory settings, where it is used as a reagent for various chemical syntheses. It serves as a source of aluminium and nitrate ions in reactions that require these components, such as in the preparation of catalysts or the synthesis of metal complexes. It is also used in analytical chemistry to test for the presence of certain substances, especially in methods that involve the precipitation of aluminium salts.
Despite its wide range of applications, aluminium nitrate nonahydrate must be handled with care due to its strong oxidizing properties. It can cause severe irritation to the skin, eyes, and respiratory system, and should be stored and used with appropriate safety precautions. In addition, its reactivity with organic materials and other reducing agents can lead to hazardous reactions, which underscores the importance of using this compound in controlled environments.
In conclusion, aluminium nitrate nonahydrate is an important chemical compound with diverse applications across industries such as explosives manufacturing, water treatment, and chemical synthesis. Its unique properties, including its ability to release aluminium and nitrate ions, make it invaluable in processes that require these components. As with many chemical substances, proper handling and safety protocols are crucial to ensuring its safe and effective use.
References
2009. Preferential adsorption of 2,4-dichlorophenoxyacetate from associated binary-solute aqueous systems by Mg/Al-NO3 layered double hydroxides with different nitrate orientations. Journal of Hazardous Materials, 165(1-3).
DOI: 10.1016/j.jhazmat.2008.10.063
2017. Silicic Acid and Beer Consumption Reverses the Metal Imbalance and the Prooxidant Status Induced by Aluminum Nitrate in Mouse Brain. Journal of Alzheimer's disease : JAD, 56(3).
DOI: 10.3233/jad-160972
2018. Can nonalcoholic beer, silicon and hops reduce the brain damage and behavioral changes induced by aluminum nitrate in young male Wistar rats? Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 118.
DOI: 10.1016/j.fct.2018.06.004
|





































































 GHS03;GHS05;GHS07 Danger Details
GHS03;GHS05;GHS07 Danger Details