Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| LTW (Jiangsu) LLC | China | |||
|---|---|---|---|---|
 | www.ltwfood.com | |||
 | +86 18505187090 | |||
 | sales9@ltwfood.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| World Metal, LLC | USA | |||
|---|---|---|---|---|
 | www.worldmetalllc.com | |||
 | +1 (877) 363-8257 (281) 491-7474 | |||
 | +1 (281) 491-7979 | |||
 | sales@worldmetalllc.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal sulfides and sulfates |
|---|---|
| Name | Manganese sulphate |
| Synonyms | Manganese (II) sulfate; Sulfuric acid manganese(2+) salt (1:1) |
| Molecular Structure | 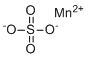 |
| Molecular Formula | MnSO4 |
| Molecular Weight | 150.99 |
| CAS Registry Number | 7785-87-7 |
| EC Number | 232-089-9 |
| SMILES | [O-]S(=O)(=O)[O-].[Mn+2] |
| Solubility | 62.9% w/w (water 20 °C) |
|---|---|
| Density | 3.25 g/mL (20 °C) |
| Melting point | 710 °C |
| Boiling point | 850 °C |
| Hazard Symbols |    GHS05;GHS08;GHS09 Danger Details GHS05;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H318-H372-H373-H411 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P270-P273-P280-P305+P354+P338-P317-P319-P391-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 1760 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Manganese sulfate, with the chemical formula MnSO4, was discovered and identified for its manganese content, a trace element essential for biological function. It consists of manganese ions (Mn²?) and sulfate ions (SO4²?) that form a soluble crystalline solid. Its solubility in water makes it useful in numerous applications ranging from agricultural to industrial processes. Manganese sulfate is primarily used as a micronutrient fertilizer in agriculture. It provides plants with essential manganese, promoting photosynthesis, enzyme activation, and overall growth. Manganese deficiency in plants can lead to reduced crop yields and poor harvest quality, so manganese sulfate is essential to correct deficiencies in manganese-deficient soils. In addition to its role as a fertilizer, manganese sulfate also acts as a soil conditioner, improving soil fertility and pH balance. It enhances plant nutrient uptake, especially in alkaline soils where manganese supplies are limited, supporting healthy plant growth and development. Manganese sulfate is used in a variety of chemical processes and industries. It is a precursor to the manufacture of other manganese compounds such as manganese dioxide, which is required for the production of dry cell batteries, ceramics, and pigments. In water treatment applications, manganese sulfate is used as a coagulant and flocculant. It helps remove turbidity and dissolved impurities from water, improving the quality of drinking water, industrial processes, and wastewater treatment. Manganese sulfate is added to animal feed as a nutritional supplement for livestock and poultry. It supports bone development, reproductive health, and overall immune function in animals, ensuring optimal growth and productivity. If used conscientiously, manganese sulfate has a minimal impact on the environment. However, its use in agricultural and industrial processes requires compliance with regulatory guidelines to prevent excessive runoff and contamination of water sources. Proper handling and disposal practices are essential to mitigate potential environmental risks. References 2024. Enhanced dual frequency microwave absorption performance of magnetic-dielectric multi-interface regulated MWCNTS/MnFe2O4/Fe3O4/Co quaternary nanocomposite. *Journal of Materials Science: Materials in Electronics*. 35(4). DOI: 10.1007/s10854-024-14006-0 2012. Tannase Production by Penicillium purpurogenum PAF6 in Solid State Fermentation of Tannin-Rich Plant Residues Following OVAT and RSM. *Applied Biochemistry and Biotechnology*. 167(5). DOI: 10.1007/s12010-012-9547-5 2005. Contribution of manganese peroxidase and laccase to dye decoloration by Trametes versicolor. *Applied Microbiology and Biotechnology*. 68(6). DOI: 10.1007/s00253-005-1964-8 |
| Market Analysis Reports |