Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sunny Industrial System GmbH | Germany | |||
|---|---|---|---|---|
 | www.sunny-gmbh.de | |||
 | +49 (211) 6999-0768 | |||
 | +49 (211) 6999-0766 | |||
 | info@sunny-gmbh.de | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Hydroxypyrimidine |
|---|---|
| Name | Isoprene |
| Synonyms | 2-Methyl-1,3-butadiene |
| Molecular Structure | 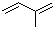 |
| Molecular Formula | C5H8 |
| Molecular Weight | 68.12 |
| CAS Registry Number | 78-79-5 |
| EC Number | 201-143-3 |
| SMILES | CC(=C)C=C |
| Density | 0.68 |
|---|---|
| Melting point | -146 °C |
| Boiling point | 34 °C |
| Refractive index | 1.4205-1.4225 |
| Flash point | -48 °C |
| Water solubility | 0.07 g/100 mL |
| Hazard Symbols |   GHS02;GHS08 Danger Details GHS02;GHS08 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H224-H350-H341-H412 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P233-P240-P241-P242-P243-P273-P280-P303+P361+P353-P318-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 1218 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Isoprene is an important organic compound with significant applications in various industries. It was discovered in the 19th century and its unique properties make it a key ingredient in the production of synthetic materials and chemicals. Isoprene (2-methyl-1,3-butadiene) was first discovered in 1860 by German chemist August Kekulé. Kekulé isolated isoprene from natural rubber and realized that its structure was a key component of polymers. The compound has since become a fundamental building block of organic chemistry. Isoprene is a colorless liquid with a characteristic odor and a boiling point of 34°C. It is highly reactive due to its conjugated double bond system, making it easy to polymerize. Its chemical formula C₅H₈ indicates that it is a diene, which means that it contains two double bonds. This reactivity allows isoprene to undergo a variety of chemical reactions, making it a very versatile compound. The most prominent application of isoprene is in the production of synthetic rubber, especially polyisoprene. Synthetic rubber is used in tires, footwear, and industrial products, mimicking the properties of natural rubber but is generally more durable and versatile. Isoprene's efficient ability to polymerize makes it an ideal precursor for these materials. Isoprene is used as a monomer in the synthesis of a variety of polymers and copolymers. It plays a role in the production of materials such as resins, adhesives, and coatings. Its reactivity allows for the creation of a variety of products with different properties, from flexible coatings to strong adhesives. Isoprene derivatives are used in the production of flavors and fragrances. Due to its unique chemical structure, the compound itself contributes to the production of certain odors and flavorings. This application leverages isoprene's role in creating complex aromatic compounds used in consumer products. In the pharmaceutical industry, isoprene is used to synthesize a variety of drugs and therapeutic agents. Its chemical versatility makes it useful in the development of compounds with specific biological activities, thereby promoting advances in medical treatments. Ongoing research focuses on exploring more sustainable and efficient methods for producing isoprene and its derivatives. Innovations in green chemistry aim to reduce environmental impact and improve the efficiency of isoprene-based processes. References 2008. Cationic borohydrido-neodymium complex: Synthesis, characterization and its application as an efficient pre-catalyst for isoprene polymerisation. Dalton Transactions, 2008(44). DOI: 10.1039/b806669h 2007. Functional analysis of genes involved in the biosynthesis of isoprene in Bacillus subtilis. Applied Microbiology and Biotechnology, 75(6). DOI: 10.1007/s00253-007-0953-5 1984. In vitro biosynthesis of isoprene from mevalonate utilizing a rat liver cytosolic fraction. Biochemical and Biophysical Research Communications, 123(2). DOI: 10.1016/0006-291x(84)90284-5 |
| Market Analysis Reports |