Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Bosche Scientific LLC, | USA | |||
|---|---|---|---|---|
 | www.boschesci.com | |||
 | +1 (732) 565-9988 | |||
 | +1 (732) 875-0899 | |||
 | sales@boschesci.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antibiotics >> Other antibiotics |
|---|---|
| Name | Cyclosporine |
| Synonyms | 30-ethyl-33-[(E,1R,2R)-1-hydroxy-2-methylhex-4-enyl]-1,4,7,10,12,15,19,25,28-nonamethyl-6,9,18,24-tetrakis(2-methylpropyl)-3,21-di(propan-2-yl)-1,4,7,10,13,16,19,22,25,28,31-undecazacyclotritriacontane-2,5,8,11,14,17,20,23,26,29,32-undecone |
| Molecular Structure | 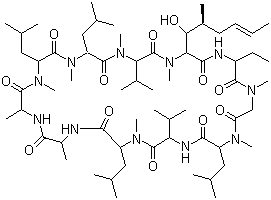 |
| Molecular Formula | C62H111N11O12 |
| Molecular Weight | 1202.61 |
| CAS Registry Number | 79217-60-0 |
| SMILES | CCC1C(=O)N(CC(=O)N(C(C(=O)NC(C(=O)N(C(C(=O)NC(C(=O)NC(C(=O)N(C(C(=O)N(C(C(=O)N(C(C(=O)N(C(C(=O)N1)[C@@H]([C@H](C)C/C=C/C)O)C)C(C)C)C)CC(C)C)C)CC(C)C)C)C)C)CC(C)C)C)C(C)C)CC(C)C)C)C |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 137 °C (Decomposes) (Expl.) |
| Solubility | Soluble (water), slightly soluble (methanol, propylene glycol) (Expl.) |
| Index of refraction | 1.475 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
Cyclosporine is a cyclic nonribosomal peptide composed of 11 amino acids, notable for its potent immunosuppressive properties. It has the molecular formula C62H111N11O12 and is produced by the fungus Tolypocladium inflatum. Discovered in the early 1970s, cyclosporine revolutionized transplant medicine by enabling organ acceptance through suppression of the immune response. Chemically, cyclosporine is a cyclic undecapeptide with a unique structure containing several unusual amino acids, including N-methylated residues, which contribute to its hydrophobic character and influence its bioavailability. This lipophilicity allows it to cross cellular membranes effectively. Cyclosporine exerts its immunosuppressive action by selectively inhibiting calcineurin, a calcium/calmodulin-dependent phosphatase critical for T-cell activation. By forming a complex with cyclophilin, an intracellular receptor, cyclosporine prevents dephosphorylation and nuclear translocation of nuclear factor of activated T-cells (NFAT), thereby suppressing the transcription of interleukin-2 and other cytokines essential for T-cell proliferation. The drug’s discovery stemmed from screening natural products for immunosuppressive activity, and it rapidly became the standard agent to prevent rejection in organ transplantation such as kidney, liver, and heart transplants. Beyond transplantation, cyclosporine is used in autoimmune diseases like psoriasis and rheumatoid arthritis, and in ophthalmology to treat dry eye disease. Pharmacokinetically, cyclosporine is characterized by variable absorption influenced by formulation and food intake. It undergoes extensive metabolism via cytochrome P450 3A enzymes in the liver and intestine, producing numerous metabolites. Its elimination half-life varies between individuals, necessitating therapeutic drug monitoring to maintain effective and safe blood concentrations. Cyclosporine’s hydrophobicity and large molecular size contribute to its complex pharmacokinetics and interactions with other drugs. It is administered orally or intravenously, with formulations designed to optimize absorption and bioavailability. Adverse effects of cyclosporine include nephrotoxicity, hypertension, neurotoxicity, and increased susceptibility to infections and malignancies due to immune suppression. Careful dose adjustment and monitoring are essential to minimize toxicity while ensuring efficacy. In summary, cyclosporine is a cyclic peptide immunosuppressant discovered from fungal sources, which inhibits T-cell activation by targeting calcineurin. Its unique chemical structure and mechanism of action have made it indispensable in organ transplantation and treatment of autoimmune conditions. References 1994. The Molecular Replacement Solution and X-ray Refinement to 2.8 Å of a Decameric Complex of Human Cyclophilin A with the Immunosuppressive Drug Cyclosporin A. Journal of Molecular Biology, 244(1). DOI: 10.1006/jmbi.1994.1738 1994. In vivo evidence for ATP-dependent and P-glycoprotein-mediated transport of cyclosporin A at the blood-brain barrier. Biochemical Pharmacology, 48(10). DOI: 10.1016/0006-2952(94)90601-7 1995. Cyclosporine in Severe Ulcerative Colitis. The New England Journal of Medicine, 332(2). DOI: 10.1056/nejm199501123320214 |
| Market Analysis Reports |