Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Qingdao Lilai Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lilaichem.com | |||
 | +86 (532) 8676-7976 | |||
 | +86 (532) 8676-7976 | |||
 | sales@lilaichem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Monomethyl sebacate |
| Synonyms | Methyl 9-carboxynonanoate; Methyl hydrogen sebacate |
| Molecular Structure | 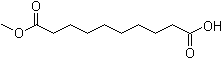 |
| Molecular Formula | C11H20O4 |
| Molecular Weight | 216.27 |
| CAS Registry Number | 818-88-2 |
| EC Number | 212-458-0 |
| SMILES | COC(=O)CCCCCCCCC(=O)O |
| Solubility | Slightly soluble (1.6 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.038±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 82 °C (ligroine )** |
| Boiling point | 205-207 °C (15 Torr)*** |
| Flash point | 115.4±16.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| ** | Zhang, Zhengbo |
| *** | Pattison, F. L. M. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Monomethyl sebacate is an organic compound derived from sebacic acid, a saturated dicarboxylic acid with a ten-carbon linear chain. Its molecular formula is C11H20O4. Structurally, monomethyl sebacate contains one methyl ester group at one terminus and a free carboxylic acid group at the other end, making it a monoester of sebacic acid. This bifunctional nature provides both ester and acid functionalities, enabling versatile chemical reactivity. Monomethyl sebacate is typically synthesized by partial esterification of sebacic acid with methanol under controlled conditions. The reaction uses acid catalysts such as sulfuric acid or p-toluenesulfonic acid, with careful monitoring to ensure esterification of only one carboxyl group, leaving the other free. Alternative methods include selective protection or activation of one acid group prior to esterification. The presence of both ester and free acid groups in monomethyl sebacate makes it a valuable intermediate in organic synthesis, polymer chemistry, and materials science. It can undergo further chemical transformations such as amidation, esterification, or coupling reactions at the free acid site, while the methyl ester can serve as a protected carboxyl group or be hydrolyzed under specific conditions. In polymer chemistry, monomethyl sebacate is used to synthesize polyesters, polyamides, and copolymers. Its bifunctionality allows incorporation as a chain terminator, modifier, or cross-linker, influencing polymer molecular weight, branching, and physical properties. The ten-carbon alkyl spacer contributes hydrophobicity and flexibility to polymer chains, affecting crystallinity and thermal behavior. Beyond polymers, monomethyl sebacate serves as a precursor for specialty chemicals, surfactants, plasticizers, and lubricant additives. Its structural features facilitate the design of amphiphilic molecules useful in detergents, emulsifiers, and personal care formulations. Physically, monomethyl sebacate is usually a colorless to pale yellow liquid or waxy solid depending on purity and temperature. It exhibits moderate solubility in organic solvents such as alcohols, ethers, and hydrocarbons, and limited solubility in water due to the hydrophobic alkyl chain. Chemically, the compound is stable under ambient conditions but may undergo hydrolysis in acidic or basic media, affecting the ester or acid groups. Proper storage in a cool, dry environment is recommended to maintain stability. Monomethyl sebacate and related sebacic acid derivatives can be obtained from renewable resources such as castor oil, supporting sustainable chemical production and green chemistry initiatives. In summary, monomethyl sebacate is a bifunctional monoester of sebacic acid containing both a methyl ester and a free carboxylic acid group. Its chemical versatility and structural properties make it a valuable intermediate in polymer synthesis, specialty chemicals, and materials science. References 2015. One-pot ozonolytic synthesis of acyclic α,ω-bifunctional compounds from methyl 10-undecenoate and 10-undecen-1-ol. Russian Journal of Applied Chemistry, 88(6). DOI: 10.1134/s1070427215060075 1998. Synthesis of polyfunctional glycerol esters: Lipase-Catalyzed esterification of glycerol with diesters. Journal of the American Oil Chemists' Society, 75(11). DOI: 10.1007/s11746-998-0092-x 1989. Chemical synthesis and spectroscopic characteristics of C18 1,2-disubstituted cyclopentyl fatty acid methyl esters. Lipids, 24(6). DOI: 10.1007/bf02535124 |
| Market Analysis Reports |