Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| The Dow Chemical Company | USA | |||
|---|---|---|---|---|
 | www.dow.com | |||
 | +1 (989) 633-1706 | |||
 | fmdcigs@dow.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Surfactant |
|---|---|
| Name | C12-C14 Secondaryalcohols ethoxylated |
| Molecular Structure | 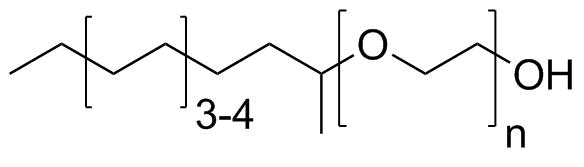 |
| CAS Registry Number | 84133-50-6 |
| EC Number | 617-534-0 |
| SMILES | CCCCCCC(C)OCCO |
| Density | 1.007 g/mL 20 $degree$C (Expl.) |
|---|---|
| Solubility | water: freely soluble (Expl.) |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H332-H315-H401 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P273-P280-P301+P312+P330-P302+P352-P304+P340+P31-P305+P351+P338+ P310-P332+P313-P362-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
C12–C14 secondary alcohols ethoxylated is a nonionic surfactant mixture derived from medium-chain (C12 to C14) secondary alcohols that have been reacted with ethylene oxide to form polyoxyethylene ether chains. It is widely used in industrial, household, and institutional cleaning formulations due to its strong wetting, emulsifying, and detergency properties. The starting material consists of secondary alcohols with 12 to 14 carbon atoms. Unlike primary alcohols, these secondary alcohols have the hydroxyl group located on an internal carbon atom rather than at the end of the chain. This structural feature typically comes from petrochemical olefin feedstocks that are hydrated or hydroformylated and then hydrogenated to yield alcohol mixtures. The resulting alcohol blend contains branched or linear isomers depending on the production route. Ethoxylation is carried out by reacting these alcohols with ethylene oxide under controlled catalytic conditions, typically in the presence of alkaline catalysts. This reaction attaches repeating ethylene oxide units (–CH2CH2O–)n to the alcohol oxygen, producing a mixture of ethoxylated homologues with varying chain lengths. The number of ethoxylate units determines the hydrophilic–lipophilic balance (HLB) and therefore strongly influences solubility, foaming, and emulsification behavior. The molecular structure consists of a hydrophobic C12–C14 alkyl chain connected through an ether linkage to a hydrophilic polyoxyethylene chain. This amphiphilic structure allows the molecule to accumulate at interfaces between oil and water, reducing surface tension and stabilizing emulsions. Because it is nonionic, it does not carry a formal charge in solution, making it compatible with a wide range of other surfactant types, including anionic, cationic, and amphoteric systems. In aqueous solution, C12–C14 secondary alcohol ethoxylates form micelles above their critical micelle concentration. These micelles can solubilize oils, grease, and hydrophobic dirt particles, enabling effective cleaning action. The ethoxylate chain interacts strongly with water through hydrogen bonding, while the alkyl chain interacts with nonpolar substances. This surfactant class is widely used in industrial and institutional cleaning products, including hard-surface cleaners, degreasers, textile processing aids, and metal cleaning formulations. It is also used in household detergents and some personal care formulations where nonionic surfactants are preferred for their mildness and stability. One of the important features of secondary alcohol ethoxylates is their relatively good detergency combined with lower foam compared to some anionic surfactants. This makes them particularly suitable for applications where controlled or low-foam cleaning is required, such as in industrial washing machines or automated cleaning systems. The ethoxylation degree can be adjusted during production to tune performance. Lower ethoxylation levels yield more hydrophobic, oil-soluble surfactants, while higher levels increase water solubility and emulsifying strength. Commercial products are typically mixtures of homologues rather than single defined molecules. From a physicochemical standpoint, these compounds exhibit typical nonionic surfactant behavior: stability in hard water, insensitivity to electrolyte concentration, and broad pH compatibility. Their performance is governed by the balance between hydrophobic alkyl chain length and hydrophilic ethoxy content. Overall, C12–C14 secondary alcohols ethoxylated are versatile nonionic surfactants derived from medium-chain secondary alcohols and ethylene oxide. Their amphiphilic structure provides effective wetting, emulsification, and detergency, making them widely used in industrial, institutional, and household cleaning formulations. References none |
| Market Analysis Reports |