Online Database of Chemicals from Around the World
| Shanghai Oripharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oripharm.com.cn | |||
 | +86 (21) 6439-6936 | |||
 | +86 (21) 6481-4386 | |||
 | info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Deyang Sinozyme Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinozyme.cn | |||
 | +86 (838) 570-2999 570-3006 | |||
 | +86 (838) 570-1388 / 570-3000 | |||
 | sinozyme@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Jiagen Biotechnologies Inc. | Canada | |||
|---|---|---|---|---|
 | www.jiagen.com | |||
 | +1 (514) 779-9623 | |||
 | info@jiagen.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Huake Labware Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shhk.com.cn | |||
 | +86 (21) 6765-2117 | |||
 | +86 (21) 5760-2161 | |||
 | shhk17@163.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Nanjing Sunsure Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunsurechem.com | |||
 | +86 (25) 5853-5435 | |||
 | +86 (25) 5853-5435 | |||
 | sales@sunsurechem.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Aajiang Jiuzhou Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuzhou-chem.com | |||
 | +86 13454675544 | |||
 | jamie@jiuzhou-chem.com | |||
 | QQ Chat | |||
 | WeChat: +86 18632988332 | |||
 | WhatsApp:+86 18632988332 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hubei Marvel-bio Medicine Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.marvel-bio.com | |||
 | +86 (027) 8773-8507 | |||
 | maoerwo123@163.com | |||
 | QQ Chat | |||
 | WeChat: kangbao813 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Lee Biosolutions, Inc. | USA | |||
|---|---|---|---|---|
 | www.leebio.com | |||
 | +1 (314) 968-1091 | |||
 | +1 (314) 968-9851 | |||
 | info@leebio.com | |||
| Chemical manufacturer | ||||
| Avanti Polar Lipids, Inc. | USA | |||
|---|---|---|---|---|
 | www.avantilipids.com | |||
 | +1 (800) 227-0651 (205) 663-2494 | |||
 | +1 (800) 229-1004 / (205) 663-0756 | |||
 | info@avantilipids.com | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Classification | Biochemical >> Enzymes and coenzymes |
|---|---|
| Name | Coenzyme A |
| Synonyms | CoA;[[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxy-3-phosphonooxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [(3R)-3-hydroxy-2,2-dimethyl-4-oxo-4-[[3-oxo-3-(2-sulfanylethylamino)propyl]amino]butyl] hydrogen phosphate |
| Molecular Structure | 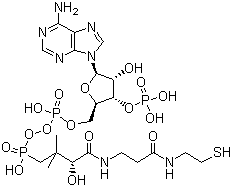 |
| Molecular Formula | C21H36N7O16P3S |
| Molecular Weight | 767.53 |
| CAS Registry Number | 85-61-0 |
| EC Number | 201-619-0 |
| SMILES | CC(C)(COP(=O)(O)OP(=O)(O)OC[C@@H]1[C@H]([C@H]([C@@H](O1)N2C=NC3=C(N=CN=C32)N)O)OP(=O)(O)O)[C@H](C(=O)NCCC(=O)NCCS)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Coenzyme A (CoA) is a key biochemical molecule that was first discovered and isolated in 1947 by Fritz Albert Lipmann while studying cellular respiration and enzyme systems. Lipmann's pioneering work revealed that CoA is an indispensable cofactor that facilitates the transfer of acyl groups in metabolic pathways, a discovery that revolutionized our understanding of biochemistry. Chemically, Coenzyme A is a small molecule composed of a pantothenic acid (vitamin B5) moiety, an adenosine diphosphate (ADP) unit, and a cysteamine group linked by a phosphodiester bond. This unique structure enables Coenzyme A to act as a carrier of acyl groups by forming a thioester bond, which is essential for the activation and transport of fatty acids and other metabolites within the cell. The primary functions of Coenzyme A revolve around its role in cellular metabolism. It is a cofactor for enzymes involved in the Krebs cycle (citric acid cycle), fatty acid oxidation, amino acid metabolism, and the biosynthesis of cholesterol, ketone bodies, and neurotransmitters. By facilitating the transfer of acyl groups, Coenzyme A plays a central role in energy production and the synthesis of biomolecules required for cellular growth and function. The significance of CoA extends beyond its metabolic role. It participates in the regulation of gene expression through the process of acetylation, affecting chromatin structure and transcriptional activity. Acetyl-CoA, a derivative of CoA, is a key substrate for histone acetyltransferases (HATs), affecting cell signaling pathways that control differentiation, proliferation, and apoptosis. In clinical medicine, CoA and its derivatives can treat diseases with impaired CoA metabolism, such as pantothenate kinase-associated neurodegeneration (PKAN), highlighting the key role of CoA in maintaining neurological function. Drug interventions targeting CoA biosynthesis or utilization pathways are under investigation for the treatment of metabolic disorders and neurodegenerative diseases. The versatility of CoA extends to biotechnological and industrial applications. Enzymatic processes utilizing CoA-dependent enzymes are used in biocatalysis to produce drugs, biofuels, and specialty chemicals. In biotechnology, CoA biosensors and CoA derivatives play an important role in metabolic engineering and synthetic biology efforts to optimize microbial production pathways. Future research on CoA will continue to explore its complex metabolic roles, regulatory functions, and potential therapeutic applications. Advances in the understanding of CoA-dependent pathways, structural biology, and enzyme engineering hold promise for the development of new therapeutics and innovative biotechnologies that harness its biochemical power. References 2024. Metabolic Function of the Adipose Tissue. Contemporary Endocrinology. DOI: 10.1007/978-3-031-72570-8_3 1991. Content of CoA-Esters in Perifused Rat Islets Stimulated by Glucose and Other Fuels. Diabetes, 40(3). DOI: 10.2337/diab.40.3.327 1991. Synthesis of myristoyl-carba(dethia)-coenzyme A and S-(3-oxohexadecyl)-coenzyme A, two potent inhibitors of myristoyl-CoA: protein N-myristoyltransferase. European Journal of Biochemistry, 195(3). DOI: 10.1111/j.1432-1033.1991.tb15756.x |
| Market Analysis Reports |