Online Database of Chemicals from Around the World
| Shandong Huimeng Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huimengchem.com | |||
 | +86 (530) 884-2888 +86 18523002917 | |||
 | +86 (530) 884-2666 | |||
 | cindy@szgoldenunion.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | 2-(2,2-Difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride |
| Molecular Structure | 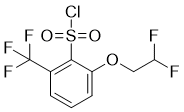 |
| Molecular Formula | C9H6ClF5O3S |
| Molecular Weight | 324.65 |
| CAS Registry Number | 865352-01-8 |
| EC Number | 881-588-3 |
| SMILES | C1=CC(=C(C(=C1)OCC(F)F)S(=O)(=O)Cl)C(F)(F)F |
| Density | 1.5±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.448, Calc.* |
| Boiling Point | 341.1±42.0 °C (760 mmHg), Calc.* |
| Flash Point | 160.1±27.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details |
|---|---|
| Risk Statements | H314 Details |
| Safety Statements | 260-P264-P280-P301+P330+P331-P303+P361+P353-P304+P340-P305+P351+P338-P310-P321-P363-P405-P501 Details |
| SDS | Available |
|
2-(2,2-Difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride is a compound that is noteworthy for its applications in the pharmaceutical and agrochemical industries. Due to its unique structure and reactivity, this compound is a key intermediate in the synthesis of various active ingredients. The development of 2-(2,2-difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride can be traced back to advances in organic synthesis aimed at creating more efficient and versatile chemical intermediates. Researchers focused on introducing fluorine atoms into organic molecules, as fluorine can significantly alter the biological activity and stability of the compounds. This led to the synthesis of this particular compound, which features a unique combination of difluoroethoxy, trifluoromethyl, and sulfonyl chloride moieties. The chemical structure of 2-(2,2-difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride includes a benzene ring substituted with 2,2-difluoroethoxy, trifluoromethyl, and sulfonyl chloride groups. This combination of substituents imparts unique chemical properties, such as increased lipophilicity and reactivity, making it a valuable intermediate in organic synthesis. While the compound itself is primarily used as an intermediate, its structure allows for a wide range of chemical reactions. The sulfonyl chloride group is highly reactive and can undergo nucleophilic substitution reactions, resulting in the formation of sulfonamide and sulfonate derivatives. These derivatives often exhibit significant biological activity, making this compound a valuable building block in drug development and agrochemical synthesis. The main application of 2-(2,2-difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride is in the pharmaceutical industry. It is used as a key intermediate in the synthesis of various therapeutic agents. For example, its derivatives can act as potent inhibitors of certain enzymes or receptors, making it useful in the development of drugs to treat diseases such as cancer, inflammation, and infectious diseases. In the agrochemical industry, this compound is used in the synthesis of herbicides and pesticides. The introduction of fluorinated groups into agrochemical compounds can enhance their efficacy and environmental stability. By modifying the structure of the active ingredient, chemists can develop products that are more effective at lower application rates, thereby reducing the environmental impact of agricultural practices. There are several advantages to using 2-(2,2-difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride in synthesis. Its high reactivity allows for the efficient production of a wide range of bioactive compounds. The presence of fluorine atoms in the molecule enhances the stability and lipophilicity of its derivatives, which can often improve the pharmacokinetic properties of drug candidates. In addition, the ability to form a variety of sulfonamide and sulfonate derivatives expands its use in medicinal chemistry and agrochemical development. Due to its reactivity, 2-(2,2-difluoroethoxy)-6-(trifluoromethyl)benzene-1-sulfonyl chloride should be handled with care. Proper safety procedures, including the use of personal protective equipment and adherence to handling guidelines, are essential to prevent exposure and ensure safe use. The compound should be stored in a cool, dry place away from moisture and incompatible materials to maintain its stability. References 2019. Convenient Synthesis of 2-(2,2-Difluoroethoxy)-6-(trifluoromethyl)-benzenesulfonyl Chloride, A Key Building Block of Penoxsulam. Synthesis, 51(20). DOI: 10.1055/s-0039-1690617 |
| Market Analysis Reports |