Online Database of Chemicals from Around the World
| Shanghai Medpep Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.medpep.com | |||
 | +86 (21) 6556-6949 | |||
 | +86 (21) 6556-9401 | |||
 | medpep@gmail.com xueminguo@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Jinxiang Chemical (Danyang) Products Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinchemical.com | |||
 | +86 (25) 8681-9862 +86 13913839793 | |||
 | +86 (25) 8681-9859 | |||
 | sales@jinchemical.com nancy-zong@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai AoBo Bio-pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aobopharm.com | |||
 | +86 (21) 5132-0130 | |||
 | +86 (21) 5132-0368 | |||
 | sales@aobopharm.com aobo@aobopharm.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shijiazhuang Haotian Chemcal Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sjzhtchem.com | |||
 | +86 (311) 8568-5134 +86 13383290929 +86 13333118931 | |||
 | +86 (311) 8568-5134 | |||
 | haotianchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Shilo Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shilopharma.com | |||
 | +86 15258872085 | |||
 | sales@shilopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Shunxiang Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunxiangchem.com | |||
 | +86 (025) 8681-9868 | |||
 | +86 (025) 8681-9859 | |||
 | sales@jinchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2025 | ||||
| AB Chem, Inc. | Canada | |||
|---|---|---|---|---|
 | www.abcheminc.com | |||
 | +1 (514) 685-8688 | |||
 | +1 (514) 685-8488 | |||
 | sales@abcheminc.com | |||
| Chemical manufacturer | ||||
| Changzhou Suntton Co., Ltd. | USA | |||
|---|---|---|---|---|
 | www.suntton.com | |||
 | +1 (914) 965-2077 +86 (519) 8513-6089 | |||
 | +1 (914) 375-2093 / +86 (519) 8513-6133 | |||
 | luke.verdet@suntton.com lily.chen@suntton.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Oxime |
|---|---|
| Name | Di-tert-Butyl azodicarboxylate |
| Synonyms | Azodicarboxylic acid di-tert-butyl ester |
| Molecular Structure | 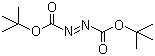 |
| Molecular Formula | C10H18N2O4 |
| Molecular Weight | 230.26 |
| CAS Registry Number | 870-50-8 |
| EC Number | 212-796-9 |
| SMILES | CC(C)(C)OC(=O)/N=N/C(=O)OC(C)(C)C |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 89 - 92 °C (Expl.) |
| Boiling point | 287.1±9.0 °C 760 mmHg (Calc.)* |
| Flash point | 107.2±13.2 °C (Calc.)* |
| Solubility | Insoluble (water) (Expl.) |
| Index of refraction | 1.46 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Di-tert-butyl azodicarboxylate (commonly abbreviated as DBAD) is an organic compound with the molecular formula C10H18N2O4. It is a symmetrical azo compound in which two tert-butyl carbamate groups (–OC(O)–) are bonded to a central azo linkage (–N=N–). The structure features two bulky tert-butyl ester substituents that confer steric hindrance and influence its chemical reactivity. DBAD is a yellow to orange crystalline solid and is typically handled as a reagent in organic synthesis. The compound is closely related in structure and reactivity to diethyl azodicarboxylate (DEAD), another well-known azo compound. Both compounds were developed and studied in the mid-20th century, with DBAD emerging as a more sterically hindered alternative to DEAD. The increased bulk of the tert-butyl groups in DBAD reduces its electrophilicity compared to DEAD, but also improves its stability and lowers its volatility, making it easier to handle and safer for certain laboratory applications. DBAD is best known for its role as an oxidizing agent and an electron-deficient azo reagent used in Mitsunobu reactions. In this context, it functions as an electron acceptor, facilitating the conversion of alcohols into esters, ethers, or other functional groups in the presence of triphenylphosphine (PPh3) and a suitable nucleophile. Although DEAD is more commonly used in Mitsunobu reactions, DBAD is sometimes preferred when lower reactivity is advantageous or when milder conditions are required. Another important use of DBAD is in cycloaddition reactions, particularly in azodicarboxylate-mediated transformations. It acts as a dienophile or electrophile in [3+2] or [4+2] cycloaddition reactions with electron-rich olefins or dipolarophiles. These reactions are often employed in the synthesis of heterocyclic compounds, such as pyrazolidines, hydrazines, or other nitrogen-containing rings. In addition, DBAD serves as a useful reagent in radical and photochemical processes. Under ultraviolet irradiation or in the presence of radical initiators, the N=N bond in DBAD can undergo homolytic cleavage, generating nitrogen-centered radicals. These reactive intermediates can participate in bond-forming reactions that are relevant in the synthesis of complex organic molecules, including pharmaceuticals and materials. The tert-butyl esters in DBAD are relatively resistant to hydrolysis, making the compound more stable toward moisture than DEAD or other lower alkyl esters. This property is beneficial during storage and handling, though DBAD should still be kept in a cool, dry place, protected from light and oxidizing agents. It is typically stored under an inert atmosphere, such as nitrogen or argon, to prevent degradation. Characterization of DBAD is achieved through standard analytical techniques. Infrared (IR) spectroscopy shows absorption bands associated with ester carbonyl stretching near 1,730 cm−1 and azo N=N stretching around 1,450–1,500 cm−1. Nuclear magnetic resonance (NMR) spectroscopy reveals signals for the tert-butyl groups and provides evidence of the symmetrical nature of the molecule. Mass spectrometry and elemental analysis are also used to confirm its molecular identity and purity. In terms of safety, DBAD, like other azodicarboxylates, should be handled with care due to its oxidative properties. Direct contact with skin or eyes should be avoided, and inhalation of dust or vapors should be minimized. Personal protective equipment, such as gloves and goggles, should be used when working with the compound in laboratory settings. In summary, di-tert-butyl azodicarboxylate is a sterically hindered azo compound used in organic synthesis as an oxidizing agent and electron-deficient reagent. Its applications include Mitsunobu reactions, cycloadditions, and radical-based transformations. With greater stability than its ethyl analogs and unique reactivity due to its bulky ester groups, DBAD serves as a valuable tool in the construction of complex molecular architectures. References 2017. Synthesis from 1,3-Dicarbonyls, Aryl Halides or Arylboronic Acids, and Di-tert-butyl Azodicarboxylate. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-112-00142 2016. From a Pyridylboronic Acid and Di-tert-butyl Azodicarboxylate. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-110-00730 2015. Amination of Arylboronic Compounds via the Copper-Catalyzed Addition of Arylboronic Esters to Azodicarboxylates. Synthesis, 47(19). DOI: 10.1055/s-0035-1560468 |
| Market Analysis Reports |