Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | tert-Butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate |
| Molecular Structure | 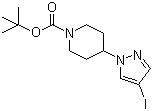 |
| Molecular Formula | C13H20IN3O2 |
| Molecular Weight | 377.22 |
| CAS Registry Number | 877399-73-0 |
| SMILES | CC(C)(C)OC(=O)N1CCC(CC1)N2C=C(C=N2)I |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
tert-Butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate is a chemical compound notable for its distinctive structure and diverse applications in organic chemistry and drug development. This substance is characterized by the presence of a tert-butyl ester group, an iodinated pyrazole moiety, and a piperidine ring. The combination of these functional groups imparts unique chemical and biological properties to the molecule, making it a subject of significant interest. The discovery of tert-butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate is rooted in the ongoing pursuit of novel compounds with potential applications in medicinal chemistry. The synthesis of this compound typically involves the introduction of the iodinated pyrazole group onto a piperidine ring, followed by esterification with tert-butyl carboxylate. This synthetic route highlights advances in chemical methodology that allow for the creation of complex molecules with precise structural features. One of the primary applications of tert-butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate is in the field of drug discovery and development. The iodinated pyrazole moiety is of particular interest due to its ability to participate in various chemical reactions and interact with biological targets. Pyrazole derivatives have been explored for their potential therapeutic effects, including anti-inflammatory, analgesic, and anticancer activities. The presence of the piperidine ring further enhances the molecule's potential as a pharmaceutical candidate by providing additional points of interaction with biological systems. The compound's structure, featuring an iodinated pyrazole group, makes it suitable for applications in radiochemistry and imaging. The iodine atom can be used as a radiotracer in imaging techniques such as positron emission tomography (PET) or single-photon emission computed tomography (SPECT). This application is particularly valuable in the development of imaging agents for diagnostic purposes, allowing researchers to track the distribution and metabolism of the compound within biological systems. In addition to its pharmaceutical and radiochemical applications, tert-butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate serves as an important intermediate in organic synthesis. The compound's ability to undergo various chemical transformations makes it a valuable building block for the synthesis of other complex molecules. Its role in constructing diverse molecular architectures underscores its utility in advancing the field of synthetic organic chemistry. Furthermore, the compound's potential extends to the development of agrochemicals. The unique combination of the pyrazole and piperidine groups could provide activity against specific pests or pathogens, making it a candidate for investigation in the creation of new pesticides or herbicides. The ability to tailor the chemical properties of the molecule through modifications to its substituents offers opportunities for designing effective agrochemical agents with improved performance. Overall, tert-butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate exemplifies the advancements in chemical synthesis and the exploration of novel compounds with diverse applications. Its discovery reflects the ongoing efforts to develop molecules with specific functionalities that can impact various fields, from pharmaceuticals to agrochemicals. References 2018. HS-1371, a novel kinase inhibitor of RIP3-mediated necroptosis. Experimental & Molecular Medicine, 50(9). DOI: 10.1038/s12276-018-0152-8 |
| Market Analysis Reports |