Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Fermentek Ltd. | Israel | |||
|---|---|---|---|---|
 | www.fermentek.com | |||
 | +972 (2) 585-3953 | |||
 | +972 (2) 585-3943 | |||
 | fermentek@fermentek.com | |||
| Chemical manufacturer since 1994 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Targetmol Chemicals Inc. | USA | |||
|---|---|---|---|---|
 | www.targetmol.com | |||
 | +1 (781) 999-5354 | |||
 | +1 (781) 281-9145 | |||
 | sales@targetmol.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2025 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Pure Research Chemicals | China | |||
|---|---|---|---|---|
 | www.purerechem.com | |||
 | +86 (551) 6288-8437 +86 18096409024 | |||
 | info@purerechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 18856022585 | |||
| Chemical manufacturer since 2018 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antifungal drugs |
|---|---|
| Name | Bafilomycin A1 |
| Synonyms | Bafilomycin A1 from Streptomyces griseus |
| Molecular Structure | 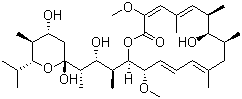 |
| Molecular Formula | C35H58O9 |
| Molecular Weight | 622.83 |
| CAS Registry Number | 88899-55-2 |
| EC Number | 621-324-4 |
| SMILES | C[C@H]1C/C(=C/C=C/[C@@H]([C@H](OC(=O)/C(=C/C(=C/[C@H]([C@H]1O)C)/C)/OC)[C@@H](C)[C@H]([C@H](C)[C@]2(C[C@H]([C@@H]([C@H](O2)C(C)C)C)O)O)O)OC)/C |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 770.1±60.0 °C 760 mmHg (Calc.)* |
| Flash point | 232.2±26.4 °C (Calc.)* |
| Solubility | Soluble DMSO 100 mM (Expl.) |
| Index of refraction | 1.535 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Bafilomycin A1 is a macrolide antibiotic that belongs to the plecomacrolide group and is primarily known as a potent and selective inhibitor of vacuolar-type H+-ATPases (V-ATPases). These enzymes are responsible for acidifying intracellular compartments such as lysosomes, endosomes, and the Golgi apparatus by transporting protons across membranes. The compound was originally isolated from *Streptomyces griseus*, and it has since become a widely used tool in cell biology for studying intracellular pH regulation and vesicular trafficking. The discovery of bafilomycin A1 dates back to the 1980s, when it was identified during screening efforts for antifungal agents. While its antifungal properties were recognized, its significance dramatically increased after its inhibitory effect on V-ATPases was characterized. By binding to the V0 domain of the V-ATPase complex, bafilomycin A1 prevents proton translocation, thereby disrupting acidification-dependent cellular processes. One of the major applications of bafilomycin A1 is in the investigation of autophagy. In autophagic pathways, V-ATPases are essential for the fusion of autophagosomes with lysosomes and the subsequent degradation of autophagic contents. Bafilomycin A1 is commonly used to inhibit the final step of autophagy, allowing researchers to distinguish between increased autophagic activity and impaired degradation. It blocks lysosomal acidification, thereby preserving autophagosome accumulation and enabling the quantification of autophagic flux when used in conjunction with other markers. In addition to autophagy research, bafilomycin A1 has been utilized to study lysosomal storage diseases, endocytic trafficking, and the intracellular survival mechanisms of certain pathogens. It has also been investigated for its role in influencing drug resistance mechanisms in cancer cells, where lysosomal sequestration and acidification can alter drug efficacy. Through its ability to increase cytoplasmic pH and prevent lysosomal acidification, bafilomycin A1 can sensitize tumor cells to various chemotherapeutic agents. The compound also influences mitochondrial function. While its primary target is the V-ATPase, studies have shown that bafilomycin A1 can also affect mitochondrial membrane potential and induce apoptosis in some contexts. These effects are believed to result indirectly from changes in intracellular ion homeostasis and energy metabolism due to disrupted lysosomal function. Despite its powerful biological activity, bafilomycin A1 is mainly used as a research reagent rather than a therapeutic agent, largely due to its broad activity and potential cytotoxicity. It lacks specificity in targeting diseased versus normal cells and can disrupt essential cellular functions. Nevertheless, its utility in experimental settings remains high due to the specificity and potency with which it inhibits V-ATPase activity. The molecular structure of bafilomycin A1 is complex, featuring a large lactone ring with multiple double bonds and side chains contributing to its biological activity. Structural variations among bafilomycin derivatives (such as A1, B1, and C1) slightly modify their biological profiles, but A1 remains the most commonly used form in laboratory research. Overall, bafilomycin A1 is a critical tool in cellular and molecular biology for studying processes reliant on acidic intracellular compartments. It has contributed to major insights into the roles of pH regulation, autophagy, vesicular transport, and intracellular signaling pathways. Its use continues to be central in exploring the mechanisms underlying cell homeostasis and disease pathology. References 1993. Bafilomycin A1 inhibits Helicobacter pylori‐induced vacuolization of HeLa cells. Molecular Microbiology, 7(2). DOI: 10.1111/j.1365-2958.1993.tb01123.x 2009. Inhibition of autophagy at a late stage enhances imatinib‐induced cytotoxicity in human malignant glioma cells. International Journal of Cancer, 124(5). DOI: 10.1002/ijc.24030 2024. Bafilomycin 1A Affects p62/SQSTM1 Autophagy Marker Protein Level and Autophagosome Puncta Formation Oppositely under Various Inflammatory Conditions in Cultured Rat Microglial Cells. International Journal of Molecular Sciences, 25(15). DOI: 10.3390/ijms25158265 |
| Market Analysis Reports |