Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shinya Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shinya-chem.cn | |||
 | +86 (316) 222-6055 | |||
 | +86 (316) 222-3178 | |||
 | shinya@shinya-chem.cn limanltw@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Reddy N Reddy Pharmaceuticals | India | |||
|---|---|---|---|---|
 | www.reddynreddypharma.com | |||
 | +91 (40) 2307-5686 +91 98480-96759 +91 98481-96759 | |||
 | info@reddynreddypharma.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Tetrahedron Scientific Inc. | China | |||
|---|---|---|---|---|
 | www.thsci.com | |||
 | +86 (571) 8848-9473 +86 18072878644 | |||
 | +86 (571) 8848-9473 | |||
 | sales@thsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Classification | Chemical reagent >> Organic reagent >> Nitro compound |
|---|---|
| Name | 4-Chloro-2-nitrotoluene |
| Synonyms | 4-chloro-1-methyl-2-nitro-benzene |
| Molecular Structure | 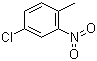 |
| Molecular Formula | C7H6ClNO2 |
| Molecular Weight | 171.58 |
| CAS Registry Number | 89-59-8 |
| EC Number | 201-921-2 |
| SMILES | CC1=C(C=C(C=C1)Cl)[N+](=O)[O-] |
| Melting point | 35-37 °C |
|---|---|
| Boiling point | 239-240 °C (718 mmHg) |
| Flash point | 120 °C |
| Water solubility | 109 mg/L (20 °C) |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335-H373-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2433; UN 3457 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
4-Chloro-2-nitrotoluene is an aromatic compound with the chemical formula C7H6ClNO2, consisting of a toluene ring substituted with a chlorine atom at the 4-position and a nitro group at the 2-position. This compound has been widely studied due to its role as an important intermediate in organic synthesis, particularly in the production of dyes, pharmaceuticals, and agrochemicals. Its reactivity, driven by the electron-withdrawing nitro and chloro groups, allows for diverse chemical transformations, making it a valuable precursor in various industrial applications. The discovery and production of 4-chloro-2-nitrotoluene were motivated by the increasing demand for versatile intermediates in the late 19th and early 20th centuries when synthetic dyes and other chemicals became critical for the textile, pharmaceutical, and agricultural industries. The compound is typically synthesized through nitration of 4-chlorotoluene, a reaction that introduces a nitro group into the aromatic ring. The process is carried out under controlled conditions, usually with a nitrating agent such as a mixture of sulfuric acid and nitric acid, to achieve the selective nitration at the desired position. One of the primary applications of 4-chloro-2-nitrotoluene is in the dye industry. Its structural framework is a useful precursor in the production of azo dyes, which are widely employed in coloring textiles, plastics, and leather. The nitro group facilitates diazotization reactions, essential for creating azo linkages that produce bright, vivid colors. Furthermore, the compound is involved in the synthesis of pigments used in various industries, ensuring the stability and vibrancy of the final products. In the pharmaceutical sector, 4-chloro-2-nitrotoluene serves as an intermediate in the synthesis of several active pharmaceutical ingredients (APIs). The nitro group is often reduced to an amino group, transforming the compound into a building block for the production of drug molecules with specific biological activities. Its role as an intermediate in creating antibacterial, antifungal, and anti-inflammatory drugs underscores its importance in medicinal chemistry. Additionally, 4-chloro-2-nitrotoluene finds application in agrochemicals, particularly in the synthesis of herbicides and pesticides. Its chemical reactivity allows for the formation of complex structures that are effective in managing pest populations and protecting crops. These agrochemical products play a crucial role in enhancing agricultural productivity and food security. Despite its industrial significance, 4-chloro-2-nitrotoluene is classified as a hazardous substance due to its potential health and environmental risks. It is known to be toxic if ingested, inhaled, or absorbed through the skin, and prolonged exposure can lead to serious health conditions. Therefore, strict safety measures are required when handling this compound to minimize risks in both industrial and laboratory environments. Overall, the discovery and application of 4-chloro-2-nitrotoluene have had a profound impact on several key industries. Its use as a precursor in dyes, pharmaceuticals, and agrochemicals highlights its versatility and importance in modern chemical manufacturing. References 2012. Molecular structure, vibrational spectra, HOMO, LUMO and NMR studies of 2-chloro-4-nitrotoluene and 4-chloro-2-nitrotoluene. Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 92. DOI: 10.1016/j.saa.2012.01.038 2009. Synthesis of 6-Substituted Imidazo[2,1-b][1,3]thiazoles and 2-Substituted Imidazo[2,1-b][1,3]benzothiazoles via Pd/Cu-Mediated Sonogashira Coupling. Synlett, 2009(15). DOI: 10.1055/s-0029-1217980 2023. Investigation of the electronic and optical activity of halogen-substituted 2-nitrotoulene by density functional theory. Optical and Quantum Electronics, 55(4). DOI: 10.1007/s11082-023-04569-3 |
| Market Analysis Reports |