Online Database of Chemicals from Around the World
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Suzhou Bichal Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bichal.com | |||
 | +86 (512) 6805-1130 | |||
 | +86 (512) 6805-2322 | |||
 | info@bichal.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Name | 4-Morpholinylacetic acid hydrochloride |
|---|---|
| Synonyms | 2-(Morpholin-4-yl)acetic acid hydrochloride |
| Molecular Structure | 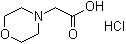 |
| Molecular Formula | C6H11NO3.HCl |
| Molecular Weight | 181.62 |
| CAS Registry Number | 89531-58-8 |
| EC Number | 675-845-7 |
| SMILES | C1COCCN1CC(=O)O.Cl |
| Melting point | 160-161 °C* |
|---|---|
| * | Brycki, Bogumil |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H318-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
4‑Morpholinylacetic acid hydrochloride (also known as morpholin‑4‑ylacetic acid HCl; CAS 89531‑58‑8) is a halogenated (salt) form of morpholine‑derived acetic acid. Its molecular formula is C6H12ClNO3 and its molar mass is approximately 181.62 g/mol. This compound is primarily used in medicinal chemistry as a pharmaceutical intermediate. Its structure features a morpholine ring (a six-membered heterocycle containing both nitrogen and oxygen) substituted with an acetic acid moiety. The protonated morpholine (from the HCl) increases the compound’s solubility in certain conditions and also stabilizes it for handling. One of the key applications of 4‑morpholinylacetic acid HCl is in peptide synthesis. It has been used in solid‑phase peptide synthesis as a building block: for example, in a study, it was incorporated via coupling (using HBTU and base) to construct a fluorogenic peptide substrate. The acid functionality allows for amide bond formation with other amino acids or peptide fragments, while the morpholine contributes to chemical stability and solubility. Another notable role is in drug‑development chemistry. According to chemical‑supplier data, it serves as an intermediate for the proteasome inhibitor **Carfilzomib**. Its morpholine-acetic acid core appears in the molecule, contributing to its pharmacokinetic and physicochemical properties. Regarding physical and safety properties, 4‑morpholinylacetic acid hydrochloride is typically obtained as a white crystalline powder of about 95 percent purity. It is reported to be insoluble in water in its salt form, which is somewhat unusual for a hydrochloride — this can influence how it is used in synthesis workflows. It should be stored in a tightly sealed container under cool, dry conditions and handled with standard lab safety precautions: it may irritate skin and eyes, and respiratory exposure should be avoided. In terms of synthesis, one documented route starts from the corresponding ester (e.g., ethyl 2‑(4‑morpholinyl)acetate), which is then refluxed in aqueous HCl. This converts the ester into the acetic acid and simultaneously forms the hydrochloride salt upon acidification. The method is relatively straightforward and scalable, making it useful for producing the intermediate in quantities suitable for further development. Overall, 4‑morpholinylacetic acid hydrochloride is a valuable chemical building block in pharmaceutical research — its combination of morpholine and carboxylic acid functionality makes it versatile for coupling, peptide chemistry, and as a point of diversity in the design of drug-like molecules. References Jia Y, Wang J, Li T, et al. (2024) Development of Inhibitors, Probes, and PROTAC Provides a Platform to Target the Atypical Kinase ERK5. Journal of Medicinal Chemistry 67(2): 856–875. DOI: 10.1021/acs.jmedchem.3c02410 Kovárí D, et al. (2025) Short scalable route to bis‑morpholine spiroacetals and spiroaminals. Journal of Organic Chemistry 90(4): DOI: 10.1021/acs.joc.4c02690 |
| Market Analysis Reports |