Online Database of Chemicals from Around the World
| Jingdezhen Fuxiang Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxiangpharm.com | |||
 | +86 (798) 833-6726 219-3329 | |||
 | +86 (798) 833-6773 | |||
 | fuxiang@fuxiangpharm.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Dawnrays Pharmaceutical (Holdings) Ltd. | China | |||
|---|---|---|---|---|
 | www.dawnrays.com | |||
 | +86 (512) 6562-6868 | |||
 | +86 (512) 6562-6968 | |||
 | gmo@dawnrays.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jinan Dechenghemu Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jndcyy.com | |||
 | +86 (531) 8888-3739 | |||
 | +86 (531) 6232-5949 | |||
 | dcyy@vip.sina.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Taizhou Jinghao Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinghaochem.com | |||
 | +86 (576) 8855-0069 +86 13486888296 | |||
 | +86 (576) 8855-7772 | |||
 | sales@jinghaochem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Wuhan Runder Pharm Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmnetcn.com | |||
 | +86 (27) 8322-9660 | |||
 | +86 (27) 8308-2226 | |||
 | runder@pharmnetcn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Zhejiang Langhua Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.langhuapharma.com | |||
 | +86 (576) 8558-8383 8558-8460 | |||
 | +86 (576) 8558-8213 / 8558-8383 | |||
 | sales@langhuapharma.com zjg@langhuapharma.com | |||
| Chemical manufacturer since 1986 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Hangzhou Cowin Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cowinpharma.com | |||
 | +86 (571) 8821-2683 8821-2056 | |||
 | +86 (571) 8821-2052 | |||
 | infor@cowinpharma.com | |||
| Chemical distributor | ||||
| Jiaojiang Xinyou Chemical Factory | China | |||
|---|---|---|---|---|
 | www.xinyouchem.com | |||
 | +86 (576) 8851-7139 | |||
 | +86 (576) 8851-7152 | |||
 | sales@xinyouchem.com ztt@xinyouchem.com | |||
| Chemical manufacturer since 1995 | ||||
| Classification | API >> Antibiotics >> Beta-lactamase inhibitor |
|---|---|
| Name | Tazobactam acid |
| Synonyms | (2S,3S,5R)-3-Methyl-4,4,7-trioxo-3-(1H-1,2,3-triazol-1-ylmethyl)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid |
| Molecular Structure | 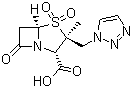 |
| Molecular Formula | C10H12N4O5S |
| Molecular Weight | 300.29 |
| CAS Registry Number | 89786-04-9 |
| EC Number | 618-303-7 |
| SMILES | C[C@@]1([C@@H](N2[C@H](S1(=O)=O)CC2=O)C(=O)O)CN3C=CN=N3 |
| Solubility | 10 mM (DMSO) (Expl.) |
|---|---|
| Density | 1.9±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.818, Calc.* |
| Boiling Point | 707.1±70.0 °C (760 mmHg), Calc.* |
| Flash Point | 381.4±35.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Dander Details GHS07;GHS08;GHS09 Dander Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H334-H373-H400-H411 Details | ||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P271-P272-P273-P280-P284-P302+P352-P304+P340-P319-P321-P333+P317-P342+P316-P362+P364-P391-P403-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Tazobactam acid is a chemical compound used primarily in the treatment of bacterial infections. It is a beta-lactamase inhibitor that is often combined with antibiotics such as piperacillin to combat infections caused by antibiotic-resistant bacteria. The discovery of tazobactam can be traced back to the 1980s, when it was developed as part of efforts to counter the increasing threat of bacterial resistance to beta-lactam antibiotics. Tazobactam acid works by inhibiting beta-lactamase enzymes, which are produced by certain bacteria to degrade beta-lactam antibiotics like penicillins and cephalosporins. By inhibiting these enzymes, tazobactam enhances the effectiveness of the co-administered antibiotics, allowing them to work more effectively against resistant strains. This mechanism of action has made tazobactam a valuable component in the treatment of infections caused by resistant pathogens, including Escherichia coli, Pseudomonas aeruginosa, and Klebsiella pneumoniae. The combination of tazobactam with piperacillin is marketed under the brand name Zosyn, and it is widely used in hospitals to treat a variety of infections, including pneumonia, intra-abdominal infections, and urinary tract infections. The effectiveness of this combination therapy has made it a standard treatment option for patients with severe infections caused by resistant organisms. Tazobactam acid itself is not used as a standalone antibiotic, as its primary role is to enhance the activity of piperacillin. In addition to its clinical uses, research into the development of newer beta-lactamase inhibitors, such as tazobactam, continues. These inhibitors play a critical role in combating the growing issue of antimicrobial resistance, and innovations in this area are essential for developing new therapies to treat difficult-to-treat infections. Tazobactam acid’s role in modern medicine highlights the importance of combating bacterial resistance, which remains one of the most significant challenges in infectious disease treatment. With ongoing advancements in research, the use of tazobactam and similar compounds will continue to evolve, offering hope for more effective treatments for resistant infections. References 2007. Biochemical Characterization of PER-2 and Genetic Environment of bla PER-2. Antimicrobial Agents and Chemotherapy, 51(7). DOI: 10.1128/aac.01395-06 2005. Quantitation and stability of piperacillin and tazobactam in plasma and ultrafiltrate from patients undergoing continuous venovenous hemofiltration by HPLC. Biomedical chromatography : BMC, 19(4). DOI: 10.1002/bmc.482 2003. Piperacillin-tazobactam is more effective than ceftriaxone plus gentamicin in febrile neutropenic patients with hematological malignancies: a randomized comparison. Supportive care in cancer : official journal of the Multinational Association of Supportive Care in Cancer, 11(6). DOI: 10.1007/s00520-003-0463-7 |
| Market Analysis Reports |