Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | API >> Blood system medication >> Anti-anemia drug |
|---|---|
| Name | Iron-dextran |
| Synonyms | Ferric hydroxide dextran complex |
| Molecular Structure | 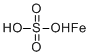 |
| Molecular Formula | FeH2O4S |
| Molecular Weight | 153.92 |
| CAS Registry Number | 9004-66-4 |
| EC Number | 233-336-3 |
| SMILES | OS(=O)(=O)O.[Fe] |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H350 Details | ||||||||||||||||||||||||||||
| Safety Statements | P202-P261-P272-P280-P302+P352-P308+P313 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Iron-dextran, a complex of iron and the polysaccharide dextran, was discovered in the mid-20th century as a solution to the challenge of delivering iron in a bioavailable form for medical purposes. The formulation was developed through the combination of iron salts with dextran, a biocompatible polymer derived from glucose. This innovation allowed for the stable encapsulation of iron, preventing its precipitation and enhancing its absorption in the body. The discovery of iron-dextran marked a significant advancement in the treatment of iron deficiency anemia, providing a safer and more effective alternative to traditional iron supplements. Iron-dextran is widely used as an intravenous therapy for iron deficiency anemia when oral iron supplements are ineffective or poorly tolerated. The formulation allows for the rapid delivery of iron directly into the bloodstream, bypassing the gastrointestinal tract and avoiding issues of poor absorption or gastrointestinal side effects. This is particularly important in cases of severe anemia or in patients with malabsorption disorders. Iron-dextran injections are often administered to pregnant women at risk of developing iron deficiency anemia. During pregnancy, the body's iron requirements increase significantly to support fetal growth and development. Iron-dextran provides a reliable means of replenishing iron stores and preventing maternal and fetal complications associated with anemia, such as preterm birth and low birth weight. Patients with chronic diseases such as chronic kidney disease (CKD) or inflammatory bowel disease (IBD) often experience iron deficiency anemia as a complication of their condition. Iron-dextran therapy is used to manage anemia in these patients, providing a steady supply of iron to support red blood cell production and improve symptoms such as fatigue and weakness. Iron-dextran injections are also utilized in veterinary medicine for the treatment of iron deficiency anemia in animals. Livestock, particularly piglets and calves, are susceptible to iron deficiency due to inadequate dietary intake or poor absorption. Iron-dextran injections help to address this deficiency and promote healthy growth and development in young animals. Iron-dextran is used in research and laboratory settings for experimental studies involving iron metabolism, anemia, and related conditions. Its well-established safety profile and consistent iron content make it a valuable tool for investigating the effects of iron supplementation and the mechanisms underlying iron deficiency and anemia. Patients undergoing hemodialysis or peritoneal dialysis are at risk of iron deficiency due to blood loss during dialysis sessions and impaired erythropoiesis. Iron-dextran injections are commonly prescribed to these patients to maintain adequate iron levels and prevent or treat anemia associated with chronic kidney disease and dialysis treatment. References 2024. Influence of an iron dextran injection in various diseases on hematological blood parameters, including serum ferritin, neonatal dairy calves. BMC Veterinary Research, 20(1). DOI: 10.1186/s12917-024-04229-y 2024. Infusion Reactions With Alternative Therapies During the National Shortage of Iron Dextran. European Journal of Haematology, 113(4). DOI: 10.1111/ejh.14322 2024. Imaging arterial and venous vessels using Iron Dextran enhanced multi‐echo 3D gradient echo MRI at 7T. NMR in Biomedicine, 37(8). DOI: 10.1002/nbm.5251 |
| Market Analysis Reports |