Online Database of Chemicals from Around the World
| Jinan Chenghui-Shuangda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnchsd.com | |||
 | +86 (531) 5889-7051 +86 15053146086 | |||
 | +86 (531) 5889-7093 | |||
 | jnchsd@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Dinghao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sddhpharm.com | |||
 | +86 (0531) 5856-5868 | |||
 | lily.zhou@sddhpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents |
|---|---|
| Name | Letermovir |
| Synonyms | (4S)-8-Fluoro-3,4-dihydro-2-[4-(3-methoxyphenyl)-1-piperazinyl]-3-[2-methoxy-5-(trifluoromethyl)phenyl]-4-quinazolineacetic acid; AIC 246 |
| Molecular Structure | 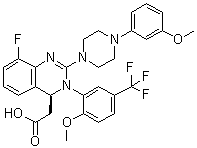 |
| Molecular Formula | C29H28F4N4O4 |
| Molecular Weight | 572.55 |
| CAS Registry Number | 917389-32-3 |
| EC Number | 805-689-9 |
| SMILES | COC1=C(C=C(C=C1)C(F)(F)F)N2[C@H](C3=C(C(=CC=C3)F)N=C2N4CCN(CC4)C5=CC(=CC=C5)OC)CC(=O)O |
| Solubility | 10 mM (DMSO) (Expl.) |
|---|---|
| Density | 1.37±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.601, Calc.* |
| Boiling Point | 706.5±70.0 °C (760 mmHg), Calc.* |
| Flash Point | 381.1±35.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Warning Details GHS08 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H361f-H373 Details | ||||||||||||
| Safety Statements | P203-P260-P273-P280-P318-P319-P391-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Letermovir is a novel antiviral drug developed for the prevention of cytomegalovirus (CMV) infection in immunocompromised patients, particularly in those undergoing stem cell or organ transplants. CMV is a common virus that can cause severe disease in individuals with weakened immune systems, such as transplant recipients or patients with HIV/AIDS. The development of letermovir was driven by the need for an effective, targeted treatment for CMV, which is known to be challenging to manage due to its ability to remain dormant in the body and reactivate when the immune system is suppressed. Letermovir is a selective inhibitor of the CMV terminase complex, which is essential for the replication of the virus. By targeting this complex, letermovir prevents the cleavage of viral DNA and inhibits the packaging of the viral genome into new virions, ultimately halting the virus's replication cycle. This mechanism of action distinguishes letermovir from other antiviral drugs, which may target different stages of the viral life cycle, such as reverse transcription or integration. Letermovir was developed by Merck & Co. and received approval from the U.S. Food and Drug Administration (FDA) in 2017 for the prevention of CMV infection in adult CMV-seropositive patients who have received an allogeneic hematopoietic stem cell transplant. Its approval was based on the results of clinical trials that demonstrated its ability to significantly reduce the incidence of CMV infection and disease in transplant recipients when compared to placebo. In addition to its use in preventing CMV infection in transplant patients, letermovir is also being investigated for potential use in other patient populations at risk for CMV infection, including solid organ transplant recipients. The drug has been shown to have a favorable safety profile, with most side effects being mild to moderate, such as gastrointestinal symptoms or mild liver enzyme elevations. The development of letermovir marks an important advancement in the field of antiviral therapies, particularly in the management of CMV infections in immunocompromised patients. Prior to the availability of letermovir, prevention of CMV relied heavily on the use of off-label drugs like ganciclovir and valganciclovir, which have more significant side effects and require more intensive monitoring. The availability of letermovir provides a safer and more effective option for preventing CMV infection in vulnerable patient populations. Letermovir represents a significant step forward in antiviral drug development, offering a more targeted and safer approach to preventing CMV-related complications in transplant recipients. As research continues, the drug may expand its indications and be utilized in additional high-risk patient populations. References 2024. Human cytomegalovirus: pathogenesis, prevention, and treatment. Molecular Biomedicine, 5(1). DOI: 10.1186/s43556-024-00226-7 2024. Impact of Letermovir for Cytomegalovirus Primary Prophylaxis on Myelosuppression and Immunosuppression in Lung Transplant Recipients. Clinical Transplantation, 38(11). DOI: 10.1111/ctr.70033 2024. Efficacy of Letermovir for Cytomegalovirus Prophylaxis Following Alemtuzumab T-Cell Depleted Allogeneic Hematopoietic Stem Cell Transplant. Transplantation and Cellular Therapy, 30(12). DOI: 10.1016/j.jtct.2024.09.009 |
| Market Analysis Reports |