Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| EAST STAR BIOTECH (SUZHOU) CO., LTD. | China | |||
|---|---|---|---|---|
 | www.eaststarbio.com | |||
 | +86 17706846989 | |||
 | daniel.creations007@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Dalian Trico Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tricochemical.com | |||
 | +86 (411) 8751-8000 8751-9854 8751-9864 | |||
 | +86 (411) 8751-9824 / 8751-9900 | |||
 | jameswang@tricochemical.com | |||
| Chemical manufacturer since 2002 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | 2-Chloroacrylonitrile |
| Synonyms | alpha-Chloroacrylonitrile |
| Molecular Structure | 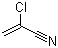 |
| Molecular Formula | C3H2ClN |
| Molecular Weight | 87.51 |
| CAS Registry Number | 920-37-6 |
| EC Number | 213-055-2 |
| SMILES | C=C(C#N)Cl |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | -65 °C |
| Index of Refraction | 1.435, Calc.* |
| Boiling Point | 89.5±13.0 °C (760 mmHg), Calc.* |
| Flash Point | 6.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software |
| Hazard Symbols |      GHS02;GHS06;GHS08;GHS05;GHS09 Danger Details GHS02;GHS06;GHS08;GHS05;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H300-H301-H310-H311-H314-H330-H334-H350-H400 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P210-P233-P240-P241-P242-P243-P260-P262-P264-P270-P271-P273-P280-P284-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P318-P320-P321-P330-P342+P316-P361+P364-P363-P370+P378-P391-P403-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3273 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2-Chloroacrylonitrile, with the chemical formula C3H2ClN, is an organic compound containing both a nitrile and a chloro group. It is typically produced by the chlorination of acrylonitrile, a process that has been known and used in industrial settings for several decades. 2-Chloroacrylonitrile is a colorless to pale yellow liquid with a pungent odor and is a reactive intermediate in organic synthesis. The primary application of 2-chloroacrylonitrile is in the synthesis of other chemicals, particularly in the production of various specialty polymers and agrochemicals. Its reactivity due to the presence of both a nitrile and a chlorine atom makes it an important intermediate in the formation of other organic compounds. In the chemical industry, 2-chloroacrylonitrile is used in the preparation of substituted acrylic compounds. These can be employed in a variety of polymerization processes, leading to the synthesis of specialty resins, coatings, and adhesives. The compound is a key intermediate in the production of acrylonitrile-butadiene-styrene (ABS) copolymers, which are widely used in engineering plastics. Its role in such applications is crucial due to its ability to form functionalized monomers that can be further polymerized to achieve desired properties. In the pharmaceutical and agricultural industries, 2-chloroacrylonitrile has been explored for its potential as a precursor to bioactive compounds. Its structure makes it suitable for modifications leading to compounds with biological activities, such as potential herbicides and fungicides. This ability to modify and synthesize new chemicals for plant protection is a key area of its use. The compound also finds its applications in the production of various intermediates used in the manufacturing of dyes, pesticides, and other fine chemicals. Its role as a building block in these sectors is vital, particularly due to its ease of functionalization and the availability of its synthetic routes. While 2-chloroacrylonitrile is useful in various industrial processes, it is also considered hazardous. It is toxic if inhaled, ingested, or absorbed through the skin. Its handling requires appropriate safety precautions to prevent exposure, and it is classified as a hazardous material under various safety regulations. In summary, 2-chloroacrylonitrile is a versatile chemical compound with applications primarily in the production of specialty chemicals, polymers, and agrochemicals. Its role as an intermediate in chemical synthesis underscores its importance in industrial chemistry. The compound’s utility in manufacturing a variety of functionalized chemicals continues to make it valuable in different sectors, though its toxicity necessitates careful handling. References 1975. Reaction of α-chloroacrylonitrile with hydrazines. Chemistry of Heterocyclic Compounds, 11(2). DOI: 10.1007/bf00471402 2013. Synthesis of bis-heterocyclic compounds with a phenylene bridge from 1,4- and 1,3-bis(2-chloro-2-cyanovinyl)benzenes. Chemistry of Heterocyclic Compounds, 49(6). DOI: 10.1007/s10593-013-1325-y 2022. Current Advances in Meerwein-type Radical Alkene Functionalizations. Synthesis, 54(22). DOI: 10.1055/s-0040-1719893 |
| Market Analysis Reports |