Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Topscience Co. Ltd. | China | |||
|---|---|---|---|---|
 | www.tsbiochem.com | |||
 | +86 (400) 820-0310 | |||
 | +86 (021) 3469-2979 | |||
 | sales@tsbiochem.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Caerulum Pharma Discovery Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.caerulumpharm.com | |||
 | +86 (21) 6455-6180 +86 189149758185 | |||
 | +86 (21) 6455-6180 | |||
 | sales-cpd@caerulumpharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridazine |
|---|---|
| Name | Resmetirom |
| Synonyms | 2-[3,5-dichloro-4-[(6-oxo-5-propan-2-yl-1H-pyridazin-3-yl)oxy]phenyl]-3,5-dioxo-1,2,4-triazine-6-carbonitrile |
| Molecular Structure | 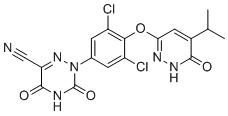 |
| Molecular Formula | C17H12Cl2N6O4 |
| Molecular Weight | 435.22 |
| CAS Registry Number | 920509-32-6 |
| SMILES | CC(C)C1=CC(=NNC1=O)OC2=C(C=C(C=C2Cl)N3C(=O)NC(=O)C(=N3)C#N)Cl |
| Solubility | 0.3519 mg/L (25 °C water) |
|---|---|
| Density | 1.7±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.725, Calc.* |
| Melting point | 349.84 °C |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Resmetirom is a selective thyroid hormone receptor-beta (THR-β) agonist developed for the treatment of nonalcoholic steatohepatitis (NASH), a chronic liver disease characterized by fat accumulation, inflammation, and liver cell damage. NASH is associated with metabolic conditions such as obesity and type 2 diabetes and can progress to cirrhosis and liver cancer if left untreated. Resmetirom was designed to target and activate the THR-β receptor specifically in the liver, promoting fat metabolism and improving liver function without the systemic effects that can occur with other thyroid hormone therapies. The discovery of resmetirom emerged from the need for more targeted therapies to manage NASH, a disease for which effective treatment options were limited. Unlike conventional thyroid hormones that act throughout the body, resmetirom selectively activates THR-β in the liver, leading to enhanced lipid metabolism, reduction of liver fat, and decreased liver inflammation. This selective activation aims to minimize the side effects typically associated with thyroid hormone treatments, such as increased heart rate and bone loss, making it a promising option for liver disease management. Clinical trials have shown that resmetirom significantly reduces liver fat content, which is a key indicator of NASH severity. In a phase 2 clinical trial, resmetirom demonstrated a notable reduction in liver fat and improvements in other biomarkers of liver health, including liver enzymes and fibrosis. These results have generated optimism for its potential to become a key therapy for NASH, a disease that currently lacks approved pharmacological treatments. Furthermore, the drug has been well-tolerated in clinical studies, with adverse effects primarily being mild and transient, such as mild gastrointestinal disturbances. In addition to its application in NASH, resmetirom is being investigated for its potential in treating other metabolic liver diseases, including conditions like liver fibrosis and non-alcoholic fatty liver disease (NAFLD). Its mechanism of action, which enhances fat oxidation and decreases fat accumulation in the liver, could prove beneficial for a range of liver diseases linked to metabolic dysfunction. Resmetirom represents a new approach to treating NASH, targeting the thyroid hormone receptor pathway to address the underlying metabolic disturbances in the liver. If future trials continue to support its efficacy and safety, resmetirom could offer a much-needed treatment option for millions of individuals suffering from NASH and other liver conditions related to metabolic diseases. References 2019. Improving NASH with a little help from thyromimetics. Lancet, 394(10213). DOI: 10.1016/s0140-6736(19)32731-x 2020. Regulation of gene transcription by thyroid hormone receptor β agonists in clinical development for the treatment of non-alcoholic steatohepatitis (NASH). PLOS ONE, 15(12). DOI: 10.1371/journal.pone.0240338 2024. Therapeutic landscape of metabolic dysfunction-associated steatohepatitis (MASH). Nature Reviews Drug Discovery, 23(12). DOI: 10.1038/s41573-024-01084-2 |
| Market Analysis Reports |