Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Dulaglutide |
| Synonyms | LY 2189265; Trulicity |
| Molecular Structure | 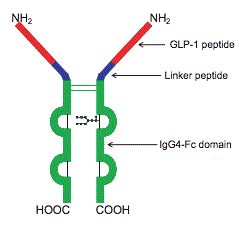 |
| Molecular Formula | C2646H4044N704O836S18 |
| Molecular Weight | 59669.81 |
| Protein Sequence | HGEGTFTSDVSSYLEEQAAKEFIAWLVKGGGGGGGSGGGGSGGGGSAESKYGPPCPPCPA$$nl$$PEAAGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSQEDPEVQFNWYVDGVEVHNAKTKP$$nl$$REEQFNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQVYTL$$nl$$PPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSRLT$$nl$$VDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLG |
| CAS Registry Number | 923950-08-7 |
|
Dulaglutide, a medication used in the management of type 2 diabetes, was developed by Eli Lilly and Company. It belongs to a class of drugs known as glucagon-like peptide-1 receptor agonists (GLP-1 RAs), which mimic the action of endogenous incretin hormones to regulate blood sugar levels. The discovery of dulaglutide stemmed from research into novel therapeutic approaches for diabetes management. Dulaglutide, a glucagon-like peptide-1 receptor agonist, is widely used in the management of type 2 diabetes mellitus. By mimicking the action of endogenous incretin hormones, dulaglutide helps regulate blood sugar levels by stimulating insulin secretion, inhibiting glucagon release, and slowing gastric emptying. It is administered via subcutaneous injection and is indicated as an adjunct to diet and exercise in patients with inadequately controlled diabetes. Clinical studies have demonstrated the efficacy of dulaglutide in reducing hemoglobin A1c levels, promoting weight loss, and lowering the risk of cardiovascular events in patients with type 2 diabetes. Additionally, dulaglutide has shown benefits in improving pancreatic beta-cell function and may offer advantages in terms of dosing frequency and tolerability compared to some other antidiabetic medications. References 2022. Once-Weekly Dulaglutide for the Treatment of Youths with Type 2 Diabetes. The New England journal of medicine, 387(5). DOI: 10.1056/nejmoa2204601 2020. The protective effects of dulaglutide against advanced glycation end products (AGEs)-induced degradation of type - collagen and aggrecan in human SW1353 chondrocytes. Chemico-Biological Interactions, 322. DOI: 10.1016/j.cbi.2020.108968 2010. Engineering and characterization of the long‐acting glucagon‐like peptide‐1 analogue LY2189265, an Fc fusion protein. Diabetes/Metabolism Research and Reviews, 26(4). DOI: 10.1002/dmrr.1080 |
| Market Analysis Reports |