Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Kingfirst Chemical (Nanjing) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingfirstchem.com | |||
 | +86 (25) 8452-2992 8477-3604 | |||
 | +86 (25) 8452-1617 | |||
 | marketing@kingfirstchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Wuhan Xiju Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | xijubio.en.made-in-china.com | |||
 | +86 13043116031 | |||
 | Fan@wh-xiju.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Z Intellecchem & Tech. Co., Ltd | China | |||
|---|---|---|---|---|
 | www.zzhxchem.com | |||
 | +86 15050866711 | |||
 | contact@zintellec.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Hubei Asli New Materials Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aslbmkpmk.com | |||
 | +86 13135651187 | |||
 | admin@aslbmkpmk.com | |||
 | Skype Chat | |||
 | WeChat: Hubei asli new materials technology co.,ltd | |||
 | WhatsApp:+86-18031176193 | |||
| Chemical distributor since 2022 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hebei Fengjia New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbfengjia.com | |||
 | +86 17133050172 | |||
 | aimee@fjchemical.com | |||
 | WhatsApp:+8617133050172 | |||
| Chemical distributor since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Organo Fine Chemicals | India | |||
|---|---|---|---|---|
 | www.organo.in | |||
 | +91 (22) 2201-6968 2206-7521 | |||
 | +91 (22) 2205-9299 / 2208-3184 | |||
 | info@organo.in | |||
| Chemical manufacturer | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Frandcom Chemicals | China | |||
|---|---|---|---|---|
 | www.fcchemicals.com | |||
 | +86 (533) 215-1679 | |||
 | +86 (533) 215-1675 | |||
 | info@frandcom.com | |||
| Chemical manufacturer since 1999 | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Hangzhou Chiral Medicine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chiralchem.com | |||
 | +86 (571) 8262-3231 8263-8910 8263-8927 | |||
 | +86 (571) 8265-4686 | |||
 | chiral@xs.hz.zj.cn | |||
| Chemical manufacturer since 1998 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Hydride, nitride, azide >> Hydride |
|---|---|
| Name | 2,5-Dimethoxybenzaldehyde |
| Synonyms | 2,5-Dimethoxy-benzaldehyde |
| Molecular Structure | 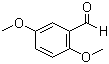 |
| Molecular Formula | C9H10O3 |
| Molecular Weight | 166.18 |
| CAS Registry Number | 93-02-7 |
| EC Number | 202-211-5 |
| SMILES | COC1=CC(=C(C=C1)OC)C=O |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 46-48 °C (Expl.) |
| Index of Refraction | 1.534, Calc.* |
| Boiling Point | 283.8 °C (760 mmHg), Calc.*, 314.4 °C (Expl.) |
| Flash Point | 120.6±8.2 °C, Calc.*, 113 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H334-H335 Details | ||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264-P264+P265-P271-P273-P280-P284-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P342+P316-P362+P364-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2,5-Dimethoxybenzaldehyde, also known as 2,5-dimethoxybenzaldehyde or simply DMB, is an organic compound with the molecular formula C8H10O3. It is a member of the benzaldehyde family, which consists of a benzene ring with a formyl group (-CHO) and additional substituent groups. Specifically, in 2,5-dimethoxybenzaldehyde, two methoxy groups (-OCH3) are attached to the benzene ring at positions 2 and 5. This compound is commonly used in the synthesis of various organic molecules and has applications in chemical research and production. The compound was first synthesized and studied in the 19th and 20th centuries, though detailed records of its discovery are relatively sparse in the literature. It is a derivative of benzaldehyde, which is known for its distinct aldehyde functional group and its use as a precursor in the synthesis of various aromatic compounds. The introduction of methoxy groups onto the aromatic ring imparts specific reactivity to the compound, making it useful for various chemical transformations and applications. One of the primary applications of 2,5-dimethoxybenzaldehyde is as an intermediate in organic synthesis. It is used in the preparation of various pharmaceutical and chemical products, including potential precursors for drugs or dyes. The methoxy groups in the molecule make it a useful building block in reactions such as electrophilic aromatic substitution, where the electron-donating effect of the methoxy groups enhances the reactivity of the aromatic ring. In the field of medicinal chemistry, 2,5-dimethoxybenzaldehyde is utilized in the synthesis of other bioactive compounds. For instance, it can serve as a precursor in the synthesis of certain psychoactive compounds, particularly in the preparation of derivatives that have been of interest in studies involving neurotransmitter modulation. Additionally, its structural features allow it to be involved in the synthesis of molecules used in cancer research, antibiotics, and anti-inflammatory agents. 2,5-Dimethoxybenzaldehyde also has applications in the synthesis of polymeric materials and specialty chemicals. The compound's ability to participate in various organic reactions allows it to be incorporated into formulations for materials that require specific properties, such as those used in coatings or advanced materials. In addition, its role as a precursor to other chemicals extends its application to the agricultural industry, where derivatives of 2,5-dimethoxybenzaldehyde can be involved in the production of pesticides or other plant protection agents. Another area where 2,5-dimethoxybenzaldehyde is used is in the field of analytical chemistry. It can be employed as a reagent in the synthesis of derivatives used for the detection and quantification of specific analytes. Its versatility in chemical reactions makes it a useful tool for researchers seeking to develop new analytical techniques or optimize existing ones. In addition to these applications, 2,5-dimethoxybenzaldehyde's use in fragrance and flavor chemistry is notable. The compound’s aromatic properties, enhanced by the methoxy groups, allow it to be utilized in the creation of complex scents or flavors. It is a key intermediate in the synthesis of various fragrances used in the cosmetic industry, as well as in flavoring agents for food and beverages. Though 2,5-dimethoxybenzaldehyde itself is not widely used as a final product in consumer goods, its role as an intermediate in synthetic chemistry makes it an important substance for the manufacture of numerous derivatives that find applications across various industries. References 2022. Corrosion performance of Schiff base derived from 2, 5-dimethoxybenzyaldehyde: X-ray structure, experimental and DFT studies. Chemical Papers, 76(9). DOI: 10.1007/s11696-022-02244-7 2020. Synthesis, characterization, investigation of mesomorphic properties and DFT studies of a new 2,5-(dimethoxy)-2-[[(4-(dodecyloxy)phenyl)imino]methyl]benzene): a material liquid crystal for optoelectronics. Journal of Materials Science: Materials in Electronics, 31(16). DOI: 10.1007/s10854-020-04145-5 2022. Synthesis of Pyrrolidine-2,3-dione Derivatives by Reacting Methyl 4-(4-Fluorophenyl)-2,4-dioxobutanoate with Tryptamine and Aromatic Aldehydes. Russian Journal of General Chemistry, 92(6). DOI: 10.1134/s1070363222060056 |
| Market Analysis Reports |