Online Database of Chemicals from Around the World
| Hangzhou King Techina Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kdqchem.com | |||
 | +86 (571) 8823-4535 | |||
 | +86 (571) 8603-9759 | |||
 | sales@kdqchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Arshine Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.arshine.com.cn | |||
 | +86 (755) 3333-0822 | |||
 | +86 (755) 8287-9323 | |||
 | marketing@arshine.com.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Rensin Chemicals Limited | China | |||
|---|---|---|---|---|
 | www.rensin-chem.com | |||
 | +86 (25) 8770-2966 | |||
 | +86 (25) 8770-2968 | |||
 | aaron@rensin-chem.com aaron.bict@hotmail.com | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Turtle Pharma Private Limited | India | |||
|---|---|---|---|---|
 | www.turtlepharma.com | |||
 | +91 9850998504 | |||
 | info@turtlepharma.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Zhejiang Langhua Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.langhuapharma.com | |||
 | +86 (576) 8558-8383 8558-8460 | |||
 | +86 (576) 8558-8213 / 8558-8383 | |||
 | sales@langhuapharma.com zjg@langhuapharma.com | |||
| Chemical manufacturer since 1986 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antibiotics >> Other antibiotics |
|---|---|
| Name | Enrofloxacin |
| Synonyms | 1-Cyclopropyl-7-(4-ethyl-1-piperazinyl)-6-fluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid |
| Molecular Structure | 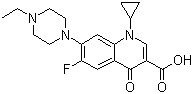 |
| Molecular Formula | C19H22FN3O3 |
| Molecular Weight | 359.40 |
| CAS Registry Number | 93106-60-6 |
| EC Number | 618-911-2 |
| SMILES | CCN1CCN(CC1)C2=C(C=C3C(=C2)N(C=C(C3=O)C(=O)O)C4CC4)F |
| Solubility | 10 mM (DMSO) |
|---|---|
| Melting point | 219-221 °C |
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H334-H361-H372-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P233-P260-P261-P264-P270-P271-P272-P273-P280-P284-P301+P317-P302+P352-P304+P340-P318-P319-P321-P330-P333+P317-P342+P316-P362+P364-P391-P403-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Enrofloxacin is a synthetic antibiotic that belongs to the fluoroquinolone class of drugs, widely used in both human and veterinary medicine. It was first developed in the 1980s by Bayer AG, a German pharmaceutical company, as part of the ongoing efforts to expand the fluoroquinolone family of antibiotics. Enrofloxacin, known for its broad-spectrum antibacterial activity, has become a valuable tool in the treatment of various bacterial infections. The discovery of enrofloxacin marked a significant advancement in antibiotic therapy. The drug is a fluorinated derivative of nalidixic acid, the first quinolone antibiotic discovered in the 1960s. The addition of a fluorine atom and other modifications in enrofloxacin resulted in enhanced antibacterial potency and a broader spectrum of activity compared to earlier quinolones. This structural refinement improved its effectiveness against a wide range of Gram-negative and Gram-positive bacteria. Enrofloxacin works by inhibiting bacterial DNA gyrase and topoisomerase IV, enzymes essential for bacterial DNA replication, transcription, and repair. By disrupting these processes, enrofloxacin prevents bacteria from reproducing, leading to their death. This mechanism of action makes enrofloxacin highly effective against various bacterial pathogens, including those resistant to other antibiotics. In human medicine, enrofloxacin is used to treat a variety of infections, including those of the respiratory tract, urinary tract, and skin. It is particularly effective against complicated infections caused by multi-drug resistant bacteria. Its broad-spectrum activity and good tissue penetration make it a valuable option in treating serious infections where other antibiotics may be less effective. Enrofloxacin's applications are particularly prominent in veterinary medicine. The drug is used extensively in the treatment of bacterial infections in companion animals such as dogs and cats, as well as in livestock. In veterinary practice, enrofloxacin is used to manage a range of conditions, including respiratory infections, urinary tract infections, and gastrointestinal infections. Its effectiveness in treating infections caused by pathogens such as Escherichia coli and Pasteurella multocida has made it a key tool in veterinary care. The drug is typically administered orally in the form of tablets or liquid formulations, and can also be given via injection in some cases. The oral formulation is particularly useful for treating a large number of animals, as it allows for easy and consistent dosing. Enrofloxacin is well-absorbed from the gastrointestinal tract and is distributed widely throughout the body, including in tissues and fluids where bacterial infections may be present. Despite its effectiveness, the use of enrofloxacin has raised concerns about the potential for developing antibiotic resistance. Bacteria can acquire resistance to fluoroquinolones through various mechanisms, including mutations in the genes encoding DNA gyrase and topoisomerase IV or increased activity of drug efflux pumps. This has led to efforts to use enrofloxacin judiciously and to explore alternative treatment options when possible. In veterinary medicine, the use of enrofloxacin is often regulated to minimize the risk of resistance development. Guidelines and best practices are established to ensure that enrofloxacin is used only when necessary and in appropriate doses, helping to preserve its effectiveness as a treatment option. In summary, enrofloxacin is a highly effective fluoroquinolone antibiotic with a broad spectrum of activity. Its discovery and development have significantly expanded the options available for treating bacterial infections in both humans and animals. However, the need for careful use and monitoring remains crucial to avoid contributing to the growing problem of antibiotic resistance. References 2008. Development of a Lateral Flow Colloidal Gold Immunoassay Strip for the Rapid Detection of Enrofloxacin Residues. Journal of Agricultural and Food Chemistry, 56(22). DOI: 10.1021/jf802648z 2005. Study of Enrofloxacin Depletion in the Eggs of Laying Hens Using Diphasic Dialysis Extraction/Purification and Determinative HPLC-MS Analysis. Journal of Agricultural and Food Chemistry, 53(7). DOI: 10.1021/jf048015u 2005. Introduction of the HPLC method for the determination of quinolone residues in various muscle tissues. Biomedical chromatography : BMC, 19(5). DOI: 10.1002/bmc.435 |
| Market Analysis Reports |