Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhongyu Medicine Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zypharmchem.com | |||
 | +86 (510) 8768-8001 | |||
 | +86 (510) 8768-6999 | |||
 | sales@zypharmchem.com jpingshi@hotmail.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Relybo Pharmachemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.relybopharma.com | |||
 | +86 (25) 8771-6566 | |||
 | susanchen@relybopharma.com | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Liyang Huibang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huibangchem.com | |||
 | +86 (519) 8784-3918 8784-3908 | |||
 | +86 (519) 8784-3928 | |||
 | sales@huibangchem.com | |||
| Chemical manufacturer | ||||
| Nata Biochemical Products Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.natachem.com | |||
 | +86 (512) 6252-0998 | |||
 | +86 (512) 6252-7116 | |||
 | natamarket@natachem.com | |||
| Chemical manufacturer since 2000 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| AB Chem, Inc. | Canada | |||
|---|---|---|---|---|
 | www.abcheminc.com | |||
 | +1 (514) 685-8688 | |||
 | +1 (514) 685-8488 | |||
 | sales@abcheminc.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Indoles |
|---|---|
| Name | 7-Methylindole |
| Synonyms | 7-Methyl-1H-indole |
| Molecular Structure | 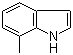 |
| Molecular Formula | C9H9N |
| Molecular Weight | 131.18 |
| CAS Registry Number | 933-67-5 |
| EC Number | 213-270-1 |
| SMILES | CC1=C2C(=CC=C1)C=CN2 |
| Water solubility | negligible (Expl.) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.* |
| Melting point | 80-84 °C (Expl.) |
| Index of Refraction | 1.655, Calc.* |
| Boiling Point | 266.0±0.0 °C (760 mmHg), Calc.*, 266 °C (Expl.) |
| Flash Point | 110.5±11.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
7-Methylindole, a derivative of indole, features a methyl group at the seventh position of the heterocyclic aromatic framework. Its molecular formula is C(_9)H(_9)N, and its structure retains the core properties of the indole nucleus, which is crucial in biochemistry, organic synthesis, and material science. The discovery of 7-methylindole can be traced back to the systematic study of indole derivatives, motivated by their biological significance and chemical versatility. Researchers aimed to explore the structural modifications of indole to investigate the impact on its chemical behavior and potential applications. Early syntheses of 7-methylindole relied on selective methylation techniques applied to the indole ring, using reagents like methyl iodide or methyl sulfate in the presence of base catalysts. In synthetic chemistry, 7-methylindole has proven to be a valuable intermediate. The methyl group enhances its reactivity in electrophilic substitution and cross-coupling reactions, facilitating the construction of complex molecular architectures. It is commonly employed in the synthesis of advanced pharmaceuticals, agrochemicals, and dyes, where the indole backbone often plays a critical role in bioactivity or functional performance. Pharmacologically, indole derivatives, including 7-methylindole, are central to the design of drugs targeting neurological and oncological pathways. The compound serves as a precursor for ligands that interact with serotonin receptors, where the indole core mimics the structure of tryptophan, a natural neurotransmitter precursor. Modifying the indole with a methyl group alters binding affinity and specificity, providing opportunities to develop selective therapeutics. In the realm of material science, 7-methylindole contributes to the development of organic semiconductors and light-emitting materials. Its conjugated structure supports charge transport and luminescence, making it a candidate for applications in organic light-emitting diodes (OLEDs) and other optoelectronic devices. Additionally, the methyl substitution enhances the thermal and chemical stability of derived materials, an essential factor in practical applications. Biologically, 7-methylindole has been studied for its antimicrobial and antifungal properties. Its derivatives have demonstrated potential as inhibitors of pathogenic organisms, suggesting uses in both clinical and agricultural settings. Moreover, the compound has been utilized as a probe in biochemical research to study the interactions of indole systems in enzymes and receptors. Further studies continue to expand the utility of 7-methylindole, focusing on its environmental applications, such as the development of green chemistry methodologies, and its role as a building block for sustainable synthetic pathways. References 2024. Unlocking therapeutic potential: exploring indole scaffolds and their structural insights as pharmacophores in designing anti-breast cancer agents. Medicinal Chemistry Research, 33(7). DOI: 10.1007/s00044-024-03241-z 2020. Differential activation of human pregnane X receptor PXR by isomeric mono-methylated indoles in intestinal and hepatic in vitro models. Toxicology Letters, 324. DOI: 10.1016/j.toxlet.2020.02.010 2016. Challenges and Opportunities in the Development of Serine Synthetic Pathway Inhibitors for Cancer Therapy. Journal of Medicinal Chemistry, 59(23). DOI: 10.1021/acs.jmedchem.6b01167 |
| Market Analysis Reports |