Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Changzhou Keylab Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.keylabchem.com | |||
 | +86 (519) 8662-2175 | |||
 | +86 (519) 8331-8678 | |||
 | wang@keylab-sh.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 3-Chloropropiophenone |
| Synonyms | 3-Chloro-1-phenyl-1-propanone; beta-Chloropropiophenone |
| Molecular Structure | 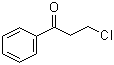 |
| Molecular Formula | C9H9ClO |
| Molecular Weight | 168.62 |
| CAS Registry Number | 936-59-4 |
| EC Number | 213-317-6 |
| SMILES | C1=CC=C(C=C1)C(=O)CCCl |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 48 - 50 °C (Expl.) |
| Boiling point | 263.3±23.0 °C 760 mmHg (Calc.)*, 301.4 - 304.3 °C (Expl.) |
| Flash point | 127.0±13.7 °C (Calc.)*, 110 °C (Expl.) |
| Solubility | Insoluble (water) (Expl.) |
| Index of refraction | 1.527 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
3-Chloropropiophenone is an organic compound belonging to the class of aryl ketones, with the molecular formula C9H9ClO. Structurally, it consists of a phenyl ring attached to a three-carbon chain bearing a ketone functional group at the first carbon and a chlorine atom substituent at the third carbon of the side chain. It is also known by the name 1-(3-chloropropyl)phenyl ketone. This compound is a colorless to pale yellow liquid with a characteristic ketonic odor and moderate solubility in organic solvents. The compound was first synthesized and characterized as part of studies exploring halogenated aryl ketones, which have been of interest due to their potential use as intermediates in pharmaceutical and agrochemical production. Its synthesis generally involves the acylation of chloropropane derivatives with benzene or substituted benzene derivatives under Friedel–Crafts acylation conditions, or via the halogenation of propiophenone at the side chain position. 3-Chloropropiophenone is primarily used as an intermediate in organic synthesis. Its functional groups—aryl ketone and alkyl chloride—offer multiple reactive sites for further chemical transformations. The chlorine atom on the propyl side chain serves as a leaving group in nucleophilic substitution reactions, allowing for the introduction of various nucleophiles to generate a wide range of derivatives, including amines, ethers, or thiols. The ketone moiety can undergo typical carbonyl chemistry such as reductions, condensations, or enolizations. In medicinal chemistry, derivatives of 3-chloropropiophenone have been investigated as precursors to bioactive compounds, including pharmaceuticals with analgesic, anti-inflammatory, or central nervous system activities. The halogenated side chain allows for modifications that can alter the lipophilicity, metabolic stability, and receptor binding properties of target molecules. Additionally, 3-chloropropiophenone has applications in agrochemical synthesis, where it can be transformed into herbicides or pesticides through further chemical modifications. The presence of both electrophilic and halogen functionalities makes it a useful scaffold for building diverse chemical entities with pesticidal activity. From an analytical perspective, 3-chloropropiophenone can be characterized by nuclear magnetic resonance (NMR) spectroscopy, which shows distinctive aromatic proton signals along with signals corresponding to the chlorinated propyl side chain and the ketone carbonyl carbon. Infrared (IR) spectroscopy confirms the presence of the carbonyl group by a strong absorption band near 1700 cm−1. Gas chromatography–mass spectrometry (GC-MS) is widely used for qualitative and quantitative analysis, especially in purity assessments and reaction monitoring. Regarding handling and safety, 3-chloropropiophenone should be stored in tightly sealed containers, away from sources of moisture and strong oxidizing agents. The compound is potentially irritating to skin, eyes, and respiratory tract, and appropriate protective measures such as gloves, goggles, and working under a fume hood are recommended during use. In summary, 3-chloropropiophenone is an important halogenated aryl ketone intermediate in organic synthesis, valued for its dual functional groups that enable a variety of chemical transformations. Its applications span pharmaceutical precursor synthesis, agrochemical development, and academic research, making it a versatile compound in industrial and laboratory settings. References 2021. Enhanced whole-cell biotransformation of 3-chloropropiophenone into 1-phenyl-1-propanone by hydrogel entrapped Chlorella emersonii (211.8b). Biotechnology Letters, 43(12). DOI: 10.1007/s10529-021-03194-y 2020. Rational design of the carbonyl reductase EbSDR8 for efficient biosynthesis of enantiopure (R)-3-chloro-1-phenyl-1-propanol. Applied Microbiology and Biotechnology, 104(24). DOI: 10.1007/s00253-020-10904-5 2015. Asymmetric hydrogenation of 3-chloro-1-phenylpropan-1-one catalyzed by ruthenium complexes. Russian Journal of General Chemistry, 85(4). DOI: 10.1134/s1070363215040271 |
| Market Analysis Reports |