Online Database of Chemicals from Around the World
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Daji Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.djpharm.com | |||
 | +86 13816245196 | |||
 | sales1@djpharm.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Biochemical >> Carbohydrate >> Oligosaccharide |
|---|---|
| Name | Ethylidene-4-nitrophenyl-a-D-Maltoheptaoside |
| Synonyms | (4aR,6R,7R,8R,8aS)-6-[(2R,3S,4R,5R,6R)-6-[(2R,3S,4R,5R,6R)-6-[(2R,3S,4R,5R,6R)-6-[(2R,3S,4R,5R,6R)-6-[(2R,3S,4R,5R,6R)-6-[(2R,3S,4R,5R,6R)-4,5-dihydroxy-2-(hydroxymethyl)-6-(4-nitrophenoxy)oxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-4,5-dihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy-2-methyl-4,4a,6,7,8,8a-hexahydropyrano[3,2-d][1,3]dioxine-7,8-diol |
| Molecular Structure | 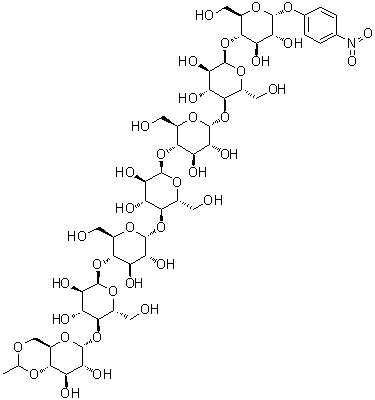 |
| Molecular Formula | C50H77NO38 |
| Molecular Weight | 1300.13 |
| CAS Registry Number | 96597-16-9 |
| SMILES | CC1OC[C@@H]2[C@@H](O1)[C@@H]([C@H]([C@H](O2)O[C@@H]3[C@H](O[C@@H]([C@@H]([C@H]3O)O)O[C@@H]4[C@H](O[C@@H]([C@@H]([C@H]4O)O)O[C@@H]5[C@H](O[C@@H]([C@@H]([C@H]5O)O)O[C@@H]6[C@H](O[C@@H]([C@@H]([C@H]6O)O)O[C@@H]7[C@H](O[C@@H]([C@@H]([C@H]7O)O)O[C@@H]8[C@H](O[C@@H]([C@@H]([C@H]8O)O)OC9=CC=C(C=C9)[N+](=O)[O-])CO)CO)CO)CO)CO)CO)O)O |
| Density | 1.8±0.1 g/cm3 Calc.* |
|---|---|
| Solubility | water: Sparingly soluble (21 g/L) (25 °C), Calc. |
| Index of refraction | 1.7 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P312+P330 Details |
| SDS | Available |
|
Ethylidene-4-nitrophenyl-α-D-maltoheptaoside is a carbohydrate derivative in which a maltoheptaose chain, composed of seven glucose units linked by α-(1→4) glycosidic bonds, is modified at the reducing end by an ethylidene group and functionalized with a 4-nitrophenyl moiety. This structural modification combines the properties of oligosaccharides with the reactivity of aromatic nitro compounds, creating a versatile molecule for enzymatic studies and synthetic applications. The 4-nitrophenyl group acts as a chromogenic leaving group, allowing the compound to serve as a substrate for glycosidase enzymes, while the ethylidene group stabilizes the reducing end, preventing undesired side reactions during chemical synthesis or enzymatic cleavage. The compound is commonly synthesized by selective glycosylation of maltoheptaose derivatives with 4-nitrophenyl ethylidene donors under controlled acidic or enzymatic conditions. Protective group strategies are often employed to ensure that glycosidic linkages remain intact and that the nitrophenyl group is attached specifically at the reducing terminus. Purification is typically achieved by column chromatography or crystallization, yielding a solid product with well-defined carbohydrate and aromatic functionality. Reaction conditions are optimized to prevent hydrolysis of glycosidic bonds or degradation of the nitrophenyl chromophore. Ethylidene-4-nitrophenyl-α-D-maltoheptaoside is primarily used as a substrate in biochemical research, particularly in the study of α-amylases, maltases, and other carbohydrate-processing enzymes. The chromogenic 4-nitrophenyl group allows quantitative monitoring of enzymatic activity by measuring the release of 4-nitrophenol, which absorbs strongly in the visible spectrum. This property makes it an invaluable tool for enzyme kinetics studies, inhibitor screening, and mechanistic investigations. By providing a well-defined oligosaccharide structure, it allows precise evaluation of enzyme specificity and substrate preference for different glycosidic linkages. In addition to enzymology, the compound has applications in synthetic carbohydrate chemistry. Its functional groups enable further derivatization, such as the introduction of additional protecting groups, conjugation to polymers, or attachment of fluorescent probes. These modifications expand the utility of ethylidene-4-nitrophenyl-α-D-maltoheptaoside as a building block for glycoarrays, biomaterials, and probes for studying carbohydrate-protein interactions. The stability of the ethylidene group ensures that the reducing end of the maltoheptaose chain remains unreactive during chemical manipulations. Physically, ethylidene-4-nitrophenyl-α-D-maltoheptaoside is typically obtained as a solid with moderate solubility in polar solvents such as dimethyl sulfoxide, dimethylformamide, and aqueous buffers. The compound is generally stable under ambient conditions but should be protected from strong acids or bases that could hydrolyze glycosidic bonds or alter the nitrophenyl chromophore. Proper storage ensures that its enzymatic and chemical reactivity are maintained for laboratory use. Overall, ethylidene-4-nitrophenyl-α-D-maltoheptaoside is a specialized carbohydrate derivative that serves as both a chromogenic substrate for enzymatic studies and a versatile intermediate in synthetic carbohydrate chemistry. Its combination of a maltoheptaose backbone, ethylidene stabilization, and 4-nitrophenyl functionality allows selective enzymatic reactions and chemical modifications, making it a valuable tool in biochemical research, glycoscience, and the development of functional oligosaccharide-based materials. References none |
| Market Analysis Reports |