Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Amino compound >> Cycloalkylamines, aromatic monoamines, aromatic polyamines and derivatives and salts |
|---|---|
| Name | Butyl methacrylate |
| Synonyms | 2-Methyl-2-propenoic acid butyl ester; 2-Methyl butyl acrylate; BMA |
| Molecular Structure | 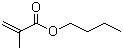 |
| Molecular Formula | C8H14O2 |
| Molecular Weight | 142.20 |
| CAS Registry Number | 97-88-1 |
| EC Number | 202-615-1 |
| SMILES | CCCCOC(=O)C(=C)C |
| Density | 0.894 |
|---|---|
| Melting point | -75 °C |
| Boiling point | 160-163 °C |
| Refractive index | 1.4225-1.4245 |
| Flash point | 41 °C |
| Water solubility | 3 g/L (20 °C) |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H315-H317-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P272-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2227 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
Butyl methacrylate is a key chemical compound extensively utilized in the production of various polymers and copolymers. It is an ester of methacrylic acid and butanol, with the molecular formula C11H18O2. This substance is highly valued for its role in enhancing the properties of plastics and coatings through polymerization. The discovery of Butyl methacrylate dates back to the early 20th century, a period marked by significant advancements in polymer chemistry. Researchers sought to develop new monomers that could improve the flexibility, clarity, and durability of synthetic materials. The incorporation of butyl groups into methacrylic acid led to the creation of Butyl methacrylate, a compound that combines the reactivity of methacrylic acid with the beneficial characteristics of butanol. In polymer chemistry, Butyl methacrylate is used primarily as a monomer in the synthesis of various copolymers. It is known for its ability to impart flexibility and impact resistance to polymers. When polymerized, Butyl methacrylate contributes to the formation of copolymers with improved mechanical properties compared to those made from methacrylic acid alone. This makes it particularly valuable in applications where materials need to withstand physical stress and retain their shape over time. One of the primary applications of Butyl methacrylate is in the production of automotive and industrial coatings. In these applications, it is used to create coatings that provide a high gloss finish, durability, and resistance to environmental factors such as UV light and moisture. The flexibility and adhesion properties imparted by Butyl methacrylate help to ensure that the coatings remain intact and effective even under harsh conditions. Butyl methacrylate is also utilized in the manufacture of adhesives and sealants. The presence of Butyl methacrylate in these formulations enhances their flexibility and bonding strength, which is crucial for applications in construction, automotive, and electronics. These adhesives and sealants are used to create strong, durable bonds between a variety of substrates, including metals, plastics, and glass. Furthermore, Butyl methacrylate is employed in the production of acrylic plastics, which are used in a wide range of consumer products, including lenses, displays, and optical devices. The addition of Butyl methacrylate to acrylic formulations improves the impact resistance and optical clarity of the resulting plastics, making them suitable for applications that require both strength and visual quality. In summary, Butyl methacrylate is a versatile and valuable compound in the field of polymer chemistry. Its discovery and subsequent applications have led to significant advancements in the performance of coatings, adhesives, and plastics. By enhancing the flexibility, durability, and clarity of materials, Butyl methacrylate continues to play a crucial role in the development of high-quality products across various industries. References 2012. The synthesis of translucent polymer nanolatexes via microemulsion polymerization. Journal of Colloid and Interface Science, 383(1). DOI: 10.1016/j.jcis.2012.06.012 2003. Polymer Brushes on Single-Walled Carbon Nanotubes by Atom Transfer Radical Polymerization of n-Butyl Methacrylate. Journal of the American Chemical Society, 125(49). DOI: 10.1021/ja037937v 2003. Complex nanostructured materials from segmented copolymers prepared by ATRP. The European physical journal. E, Soft matter, 12(1). DOI: 10.1140/epje/e2003-00009-x |
| Market Analysis Reports |