Online Database of Chemicals from Around the World
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluorotoluene series |
|---|---|
| Name | 4-Chlorobenzotrifluoride |
| Synonyms | 1-chloro-4-(trifluoromethyl)benzene; 4-Chloro-alpha,alpha,alpha-trifluorotoluene; (p-Chlorophenyl)trifluoromethane; p-Chlorotrifluoromethylbenzene; p-(Trifluoromethyl)chlorobenzene; PCBTF |
| Molecular Structure | 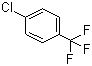 |
| Molecular Formula | C7H4ClF3 |
| Molecular Weight | 180.55 |
| CAS Registry Number | 98-56-6 |
| EC Number | 202-681-1 |
| SMILES | C1=CC(=CC=C1C(F)(F)F)Cl |
| Density | 1.353 |
|---|---|
| Melting point | -36 °C |
| Boiling point | 136-138 °C |
| Refractive index | 1.4453-1.4473 |
| Flash point | 47 °C |
| Water solubility | 29 ppm (23 °C) |
| Hazard Symbols |    GHS02;GHS07;GHS09 Warning Details GHS02;GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H315-H317-H319-H335-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P272-P273-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P370+P378-P391-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2234 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
4-Chlorobenzotrifluoride (CBTF) is an aromatic organic compound characterized by the presence of a chlorobenzene moiety with three fluorine substituents. It is a colorless to pale yellow liquid that is primarily used as a solvent and reagent in various chemical processes. The discovery of CBTF dates back to the late 20th century when researchers began exploring the utility of chlorinated and fluorinated compounds in industrial applications. Its unique chemical properties make it a valuable compound in organic synthesis and material science. The synthesis of 4-chlorobenzotrifluoride typically involves the halogenation of chlorobenzene using trifluoroacetic acid or through the reaction of chlorobenzene with trifluoromethyl iodide in the presence of a suitable catalyst. This synthetic pathway allows for the introduction of the trifluoromethyl group, which enhances the compound's stability and solubility in various organic solvents. The versatility of the synthesis methods has facilitated its use in both laboratory and industrial settings. One of the primary applications of 4-chlorobenzotrifluoride is as a solvent in organic synthesis. Its high polarity and ability to dissolve a wide range of organic compounds make it an ideal medium for various chemical reactions, including nucleophilic substitutions and polymerizations. CBTF is especially useful in reactions involving sensitive functional groups, as it minimizes unwanted side reactions that may occur in less selective solvents. In addition to its role as a solvent, 4-chlorobenzotrifluoride serves as an important reagent in the synthesis of pharmaceutical intermediates and agrochemicals. Its trifluoromethyl group is a key feature in many biologically active molecules, providing enhanced lipophilicity and metabolic stability. The incorporation of CBTF into drug development processes has led to the creation of novel compounds with improved therapeutic profiles. Another notable application of 4-chlorobenzotrifluoride is in the field of polymer chemistry. CBTF is used as a monomer in the production of fluorinated polymers, which exhibit excellent chemical resistance and thermal stability. These polymers are employed in various industries, including electronics, aerospace, and automotive, where durability and performance are critical. Moreover, the unique properties of 4-chlorobenzotrifluoride have attracted interest in the field of materials science. It can be utilized in the preparation of advanced materials such as coatings, adhesives, and sealants, where its chemical stability and compatibility with other materials enhance product performance. The ability to tailor the physical and chemical properties of materials through the incorporation of CBTF opens new possibilities for innovative applications. Despite its advantages, safety considerations surrounding the use of 4-chlorobenzotrifluoride are important. As with many halogenated compounds, it is essential to handle CBTF with care due to its potential environmental impact and health risks. Regulatory assessments and guidelines are in place to ensure safe handling, storage, and disposal of the compound in industrial and laboratory settings. Research into 4-chlorobenzotrifluoride continues, focusing on its potential applications in green chemistry and sustainable processes. The ability to utilize CBTF in reactions that minimize waste and improve reaction efficiency aligns with the growing demand for environmentally friendly chemical processes. In conclusion, 4-chlorobenzotrifluoride is a versatile compound with significant applications in organic synthesis, polymer chemistry, and materials science. Its unique chemical properties and synthetic versatility make it a valuable tool in the development of novel compounds and materials. As research progresses, the potential for 4-chlorobenzotrifluoride in new applications continues to expand, reinforcing its importance in modern chemistry. References 2024. The iron-catalysed Suzuki coupling of aryl chlorides. Nature Catalysis, 7(10). DOI: 10.1038/s41929-024-01234-0 2023. Rapid reaction optimization by robust and economical quantitative benchtop 19F NMR spectroscopy. Nature Protocols, 19(2). DOI: 10.1038/s41596-023-00951-3 2022. A Review on the Halodefluorination of Aliphatic Fluorides. Synthesis, 54(1). DOI: 10.1055/a-1684-0121 |
| Market Analysis Reports |