Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai Conson Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.consonchem.com | |||
 | +86 (21) 6354-8358 | |||
 | +86 (21) 6353-6992 | |||
 | sales@consonchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Xuzhou Ruisai Technology Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.risachem.com | |||
 | +86 (526) 8776-8018 | |||
 | +86 (526) 8776-8008 | |||
 | sales@risachem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Wuhan Ruiji Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruijichemical.com | |||
 | +86 (27) 8325-6046 +86 13871221857 | |||
 | +86 (27) 5135-8498 | |||
 | sales@ruijichemical.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| SLN Pharmachem | India | |||
|---|---|---|---|---|
 | www.slnpharmachem.com | |||
 | +91 (22) 2870-1926 6524-0421 2854-9125 | |||
 | +91 (22) 2854-9125 | |||
 | ho@slnpharmachem.com office@slnpharmachem.co.in | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Mesochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mesochem.com | |||
 | +86 (10) 5786-2036 57862181 67374028 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | sales@mesochem.com huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Changzhou Jintan ChengEn Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chengenchem.com | |||
 | +86 (519) 8019-0155 +86 13961176135 +86 13775159780 | |||
 | +86 (519) 8019-0155 | |||
 | sales@chengenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Jinkui(taizhou)technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jktland.com | |||
 | +86 (576) 8846-3883 | |||
 | chowsale001@hotmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 3'-Aminoacetophenone |
| Synonyms | m-Aminoacetophenone; 1-(3-Aminophenyl)ethanone |
| Molecular Structure | 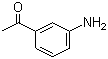 |
| Molecular Formula | C8H9NO |
| Molecular Weight | 135.16 |
| CAS Registry Number | 99-03-6 |
| EC Number | 202-722-3 |
| SMILES | CC(=O)C1=CC(=CC=C1)N |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 94-98 °C (Expl.) |
| Index of Refraction | 1.571, Calc.* |
| Boiling Point | 290.3±13.0 °C (760 mmHg), Calc.*, 289-290 °C (Expl.) |
| Flash Point | 129.4±19.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319 Details | ||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P305+P351+P338-P330-P337+P317-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3'-Aminoacetophenone is an organic compound belonging to the class of aromatic amines. It consists of a phenyl group attached to a carbonyl group (acetophenone) with an amino group (-NH2) at the 3' position of the aromatic ring. This molecule is used as an intermediate in organic synthesis, especially in the production of various bioactive compounds, pharmaceuticals, and agrochemicals. The discovery and development of 3'-aminoacetophenone fall within the larger context of acetophenone derivatives. Acetophenone, which is itself a simple aromatic ketone, has been widely used as a starting material in organic chemistry for the synthesis of more complex molecules. The introduction of functional groups like amino and carbonyl enhances the reactivity of the molecule and opens up possibilities for further chemical transformations. 3'-Aminoacetophenone can be synthesized through various synthetic routes, typically involving the functionalization of acetophenone with an amine group. One common method involves the nitration of acetophenone to produce a nitroacetophenone derivative, which can then be reduced to the corresponding aminoacetophenone. Another synthetic approach includes the direct introduction of an amino group at the 3' position via electrophilic aromatic substitution reactions under suitable conditions. The primary applications of 3'-aminoacetophenone are in the field of medicinal chemistry and materials science. In medicinal chemistry, it serves as a key intermediate for the synthesis of various bioactive molecules. It has been explored in the design and development of pharmaceutical agents, particularly those with antibacterial, anti-inflammatory, or anticancer properties. The amino group at the 3' position allows for the modification of the molecule to enhance its biological activity, such as improving its solubility or its ability to interact with specific biological targets. Moreover, 3'-aminoacetophenone has been used in the preparation of compounds with potential neuroactive properties. The acetophenone scaffold, along with the amino group, provides a structural base for molecules that can interact with the central nervous system, and these molecules have been studied for their ability to modulate neurotransmitter activity. In addition to its applications in pharmaceuticals, 3'-aminoacetophenone plays a role in materials science. Its structural features make it useful in the synthesis of organic semiconductors and other advanced materials, including organic light-emitting diodes (OLEDs) and organic solar cells. The ability to modify the molecule through further substitution or functionalization allows for tuning the electronic properties of the material, making it suitable for use in various electronic and optoelectronic devices. In summary, 3'-aminoacetophenone is an important intermediate in organic synthesis with established applications in pharmaceuticals, materials science, and the development of bioactive molecules. Its ability to serve as a building block for more complex compounds has made it a valuable substance in both research and industrial applications. References 1985. Reaction of substituted acetophenones with pentafluorophenyltrimethylsilane and related compounds under conditions of nucleophilic catalysis. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 34(11). DOI: 10.1007/bf00956809 2018. Mono- and dinuclear palladium(II) complexes incorporating 1,2,3-triazole-derived mesoionic carbenes: syntheses, solid-state structures and catalytic applications. Transition Metal Chemistry, 43(8). DOI: 10.1007/s11243-018-0267-8 2021. Theoretical and experimental approaches of new Schiff bases: efficient synthesis, X-ray structures, DFT, molecular modeling and ADMET studies. Journal of the Iranian Chemical Society, 18(10). DOI: 10.1007/s13738-021-02194-z |
| Market Analysis Reports |