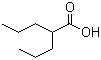
2-Propylpentanoic acid, commonly known as valproic acid, is a carboxylic acid derivative that was first synthesized in the late 19th century. Initially, it was used as an organic solvent due to its lipophilic properties. However, its pharmacological potential was discovered in the 1960s when researchers observed its anticonvulsant activity. Since then, valproic acid has been widely used in medicine, primarily for the treatment of epilepsy and other neurological disorders.
Valproic acid acts by increasing the availability of gamma-aminobutyric acid (GABA), an inhibitory neurotransmitter in the central nervous system. This effect helps to stabilize neuronal activity, making it effective in controlling seizures. In addition to its role as an anticonvulsant, valproic acid has mood-stabilizing properties and is commonly prescribed for bipolar disorder. It is also used as a preventive treatment for migraine headaches.
Valproic acid is available in various formulations, including sodium valproate and divalproex sodium, which differ in their pharmacokinetic profiles but share the same therapeutic effects. The drug is well absorbed following oral administration, with metabolism occurring primarily in the liver through glucuronidation and mitochondrial β-oxidation. Due to its hepatic metabolism, interactions with other drugs that affect liver enzymes can influence its plasma levels and efficacy.
In clinical use, valproic acid is generally well tolerated, though it has known adverse effects, including gastrointestinal disturbances, weight gain, tremors, and hepatotoxicity in rare cases. Its use during pregnancy is associated with an increased risk of teratogenic effects, particularly neural tube defects, which has led to strict guidelines regarding its administration in women of childbearing potential.
Beyond its established applications in neurology and psychiatry, valproic acid has been investigated for its potential effects in other medical conditions, including neurodegenerative diseases and certain types of cancer. However, its primary role remains in the management of epilepsy and bipolar disorder, where it continues to be a key therapeutic agent.
References
2025. DNA methylation profiles of transgenerational rat hyperactivity primed by silver nanoparticles: Comparison with valproate model rats of autism. Behavioural Brain Research, 477.
DOI: 10.1016/j.bbr.2024.115293
2025. Valproic acid-induced oxidative stress: Systematic review, meta-analysis and network pharmacology highlights disruption in antioxidant pathways in rodents. Toxicology and Applied Pharmacology, 484.
DOI: 10.1016/j.taap.2024.117160
2024. Glutamic Acid Decarboxylase 65 Antibody-associated Epilepsy and Diplopia: Two Case Reports with Literature Review. Cerebellum (London, England), 23(6).
DOI: 10.1007/s12311-024-01768-w
|



































 GHS05;GHS07;GHS08 Danger Details
GHS05;GHS07;GHS08 Danger Details