Online Database of Chemicals from Around the World
| Changzhou Chenqiang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cq-chem.com | |||
 | +86 (519) 8890-2455 (510) 6689-6080 +86 18015336699 | |||
 | +86 (519) 8890-4599 | |||
 | info@cq-chem.com sales@wxichem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Nanjing Hike Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njhkchem.com | |||
 | +86 (25) 5211-0956 | |||
 | +86 (25) 5211-0956 | |||
 | sophia@njhkchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17372769668 | |||
 | WhatsApp:17372769668 | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Thermo Fisher Scientific | USA | |||
|---|---|---|---|---|
 | www.fishersci.com | |||
 | +1 (800) 766-7000 | |||
 | +1 (800) 926-1166 | |||
 | fs.order@thermofisher.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon nitrite |
|---|---|
| Name | Ethyl p-nitrobenzoate |
| Synonyms | Ethyl 4-nitrobenzoate; 4-Nitrobenzoic acid ethyl ester |
| Molecular Structure | 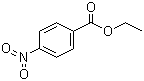 |
| Molecular Formula | C9H9NO4 |
| Molecular Weight | 195.17 |
| CAS Registry Number | 99-77-4 |
| EC Number | 202-786-2 |
| SMILES | CCOC(=O)C1=CC=C(C=C1)[N+](=O)[O-] |
| Melting point | 56-59 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Ethyl p-nitrobenzoate, an organic compound with the formula C9H9NO4, was first synthesized in the late 19th century during investigations into nitration reactions of aromatic esters. This compound is typically produced by the esterification of p-nitrobenzoic acid with ethanol in the presence of acid catalysts. The discovery of ethyl p-nitrobenzoate provided a useful synthetic intermediate for various organic chemistry applications. Ethyl p-nitrobenzoate serves as a versatile intermediate in organic synthesis. Its structure, which includes both an ester and a nitro group, makes it reactive and suitable for various chemical transformations. The nitro group can be reduced to an amino group, producing ethyl p-aminobenzoate, a precursor for many dyes, pigments, and pharmaceuticals. The ester group can undergo hydrolysis, yielding p-nitrobenzoic acid, which can then be used in further syntheses. In the pharmaceutical industry, ethyl p-nitrobenzoate is used to synthesize active pharmaceutical ingredients (APIs) and intermediates. The nitro group can be transformed into various functional groups, allowing the design of molecules with specific biological activities. Ethyl p-nitrobenzoate is employed in the agrochemical sector to produce herbicides, insecticides, and fungicides. The compound's chemical structure allows for the formulation of agrochemicals with specific modes of action, targeting particular pests or pathogens while minimizing harm to the crops. In materials science, ethyl p-nitrobenzoate is used in the development of advanced materials, including polymers and resins. Its reactivity with various monomers allows the creation of polymers with unique properties, such as enhanced thermal stability and chemical resistance. These materials are used in coatings, adhesives, and other applications where performance and durability are critical. The compound's ability to introduce functional groups into polymer chains makes it a useful component in developing high-performance materials. Ethyl p-nitrobenzoate is also used in the dye and pigment industry. Its derivatives can be used to produce dyes with vibrant colors and excellent fastness properties, suitable for textiles, inks, and coatings. In academic and industrial research, ethyl p-nitrobenzoate is utilized to explore new synthetic methodologies and reaction mechanisms. Researchers use it as a model compound to study nitration, esterification, and reduction reactions. References 2020. Two step continuous-flow synthesis of benzocaine. Journal of Flow Chemistry, 10(3). DOI: 10.1007/s41981-020-00098-2 2019. Palladium Supported on Carbon Nanoglobules as a Promising Catalyst for Selective Hydrogenation of Nitroarenes. Catalysis Letters, 149(12). DOI: 10.1007/s10562-019-02974-6 2007. Scandium Triflate Catalyzed Transesterification of Carboxylic Esters. Synlett, 2007(2). DOI: 10.1055/s-2007-967936 |
| Market Analysis Reports |